
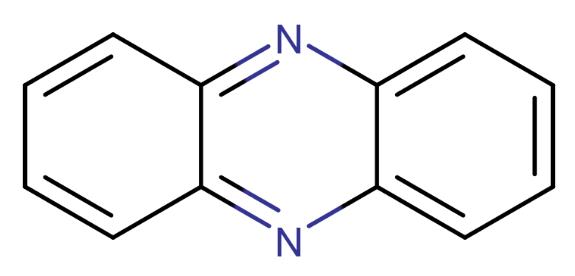
Phenazine is a class of nitrogen-containing heterocyclic compounds that are either natural or synthetic. Natural phenazines are mainly found in marine and terrestrial microorganisms. To date, over 100 natural phenazine derivatives and more than 6,000 synthetic phenazine derivatives have been studied. Phenazine derivatives are a large group of planar nitrogen heterocyclic compounds, with the most important core structure being a pyrazine ring (1,4-diazabenzene) with two ringed benzene groups. The chemical and physical properties of phenazine derivatives vary due to the type and location of functional groups. Their redox and fluorescent properties have attracted increasing attention. Some are used as important dyes in medicine and the biological industry, while others have been developed as effective fluorescent probes to study biochemical changes within the body.
Phenazine and its derivatives were initially used in the dye industry but were later discovered to have significant biological activity and can act as antimicrobial agents. In recent years, the application of phenazine has expanded to include pharmaceuticals, pesticides, chromophores, conductors, and battery materials.
Research has shown that 5,10-N,N-dihydroxyphenazine and its related compounds exhibit the strongest activity against certain microorganisms, while 1-hydroxyphenazine and safranine (a basic derivative of phenazine) also demonstrate significant antimicrobial effects. Phenazine derivatives have the potential to treat diseases caused by drug-resistant microorganisms. Phenazine compounds include phenazine-N-oxides:
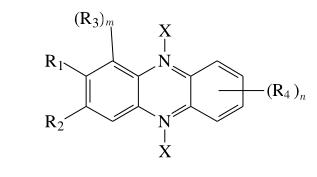
Notably, phenazine-5,10-dioxide and 7,8-substituted phenazine-5,10-dioxides. Drug-resistant bacteria and viruses include Hemophilus spp, E. coli, Enterobacter spp, Citrobacter spp, Proteus spp, Morganella spp, Shigella spp, Yersinia spp, Salmonella spp, and others.
Cardozo et al. studied an important phenazine metabolite for its antibacterial activity against MRSA (methicillin-resistant Staphylococcus aureus) strains, finding that it exhibits synergistic effects when combined with silver nanoparticles produced by Fusarium species. Thanabalasingam et al. first isolated it from the leaves of the medicinal plant Red Poppy. Tupe et al. tested its activity against human pathogen Candida albicans (MIC = 32-64 μg/mL), demonstrating its antimicrobial and antifungal effects through a mechanism mediated by reactive oxygen species (ROS); it leads to ROS production within cells. ROS causes mitochondrial membrane hyperpolarization, followed by phosphatidylserine externalization, chromatin condensation, and DNA fragmentation, inducing apoptosis and ultimately cell death. Kennedy et al. and Ali et al. further explored its anticancer mechanisms. The anticancer activity mechanism is also related to ROS. Overexpression of p53, Bax, and cytochrome C (Cyto-C) occurs, while apoptosis and anti-apoptotic proteins such as poly(ADP-ribose) polymerase (PARP) and B-cell lymphoma-2 (Bcl-2) family proteins (Bcl-2, Bcl-w, and Bcl-xL) are suppressed.
Phenazine-11-carboxamide derivatives:
![Benzo[a]phenazine-11-amide derivatives: Benzo[a]phenazine-11-amide derivatives:](https://imgen4.guidechem.com/img/images/2024/9/13/172622137741878.jpg)
These can act as dual inhibitors of topoisomerases I and II, showing potential for treating various cancers, including leukemia, lymphoma, sarcoma, and cancers of the breast, colon, brain, lung, ovary, pancreas, stomach, and skin. Additionally, it has antiviral, antibacterial, and antifungal properties. The drug can be administered in various forms, including oral tablets, capsules, suspensions, and through intramuscular, intravenous, or subcutaneous injections.
One phenazine product is promazine, used to treat nausea, vomiting, allergic reactions, and motion sickness. It is also used for short-term treatment of adult insomnia (difficulty sleeping) and as a sedative for children aged five and older. Possible side effects of using phenazine include dry mouth, blurred vision, dizziness, drowsiness, nausea, sedation, tremors, seizures, hypertension, hypotension, asthma, nasal congestion, respiratory depression, apnea, pain at the injection site, swelling, redness, tissue necrosis, and gangrene (tissue death caused by infection or inadequate blood flow).
Phenazine dyes, particularly intermediates in phenazine-based colorant dyes, have been used in industry for over 100 years. The earliest dye known as safranine (1-hydroxy-5-methylphenazine, 1), with the following structure:
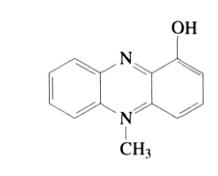
Notable phenazine dyes include Heliotrope B (bloodstone B) and Phenosafranine (phenol-safranine). All phenazine dyes that provide red, orange, and magenta hues have an unsubstituted amino group at the 3-position, 7-position, or both the 3,7-positions:
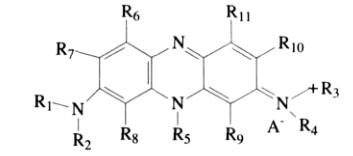
Phenazine test papers are used to measure pH, providing distinct and clear changes within a pH range of 4.5 to 7.5.
Phenazine and its derivatives are important fine chemical raw materials with widespread uses in pharmaceuticals, pesticides, dyes, organic conductors, and solar cells. Recently, organic solid-state light-emitting diodes have attracted considerable attention as potential low-cost solar cells, though current organic photovoltaic cells are much less efficient than silicon semiconductor photovoltaic cells. Takeshi et al. proposed the concept of D-σ-A molecules/conductive polymer photovoltaic cells and successfully fabricated a gold-[poly(3-methylthiophene)]-[10-(p-nitrobenzyl)-2-(10H)phenazine ketone](25)-aluminum photovoltaic cell, achieving an energy conversion efficiency of 0.051%. This result demonstrates its potential as a low-cost solar cell. With further research, the application of phenazine derivatives in emerging fields like energy is expected to see broader development.
[6] Yan J, Liu W, Cai J, et al. Advances in Phenazines Over the Past Decade: Review of Their Pharmacological Activities, Mechanisms of Action, Biosynthetic Pathways and Synthetic Strategies [J]. Marine Drugs, 2021, 19(11): 610.
 |
 |
 |