-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

Iodomethane, with a chemical formula CH3I, is a dense, colorless, and volatile liquid. Structurally similar to methane, it substitutes one hydrogen atom with an iodine atom. Natural emissions of iodomethane occur from rice paddies. Additionally, marine algae and seaweed in temperate oceans produce over 214,000 tons of iodomethane annually. Trace amounts are also emitted by fungi and bacteria on land. In organic synthesis, iodomethane serves as a source of methyl groups.
Iodomethane (CH3I), with iodomethane molecular weight of 141.94, is formed by replacing one hydrogen atom in methane with an iodine atom. It adopts a simple tetrahedral structure where a carbon atom (C) bonds covalently with three hydrogen atoms (H) and one iodine atom (I). Due to the size difference between atoms, the carbon-iodine bond is the longest and weakest in this structure, influencing iodomethane's reactivity and susceptibility to cleavage in various chemical reactions. The structural formula of iodomethane is as follows:
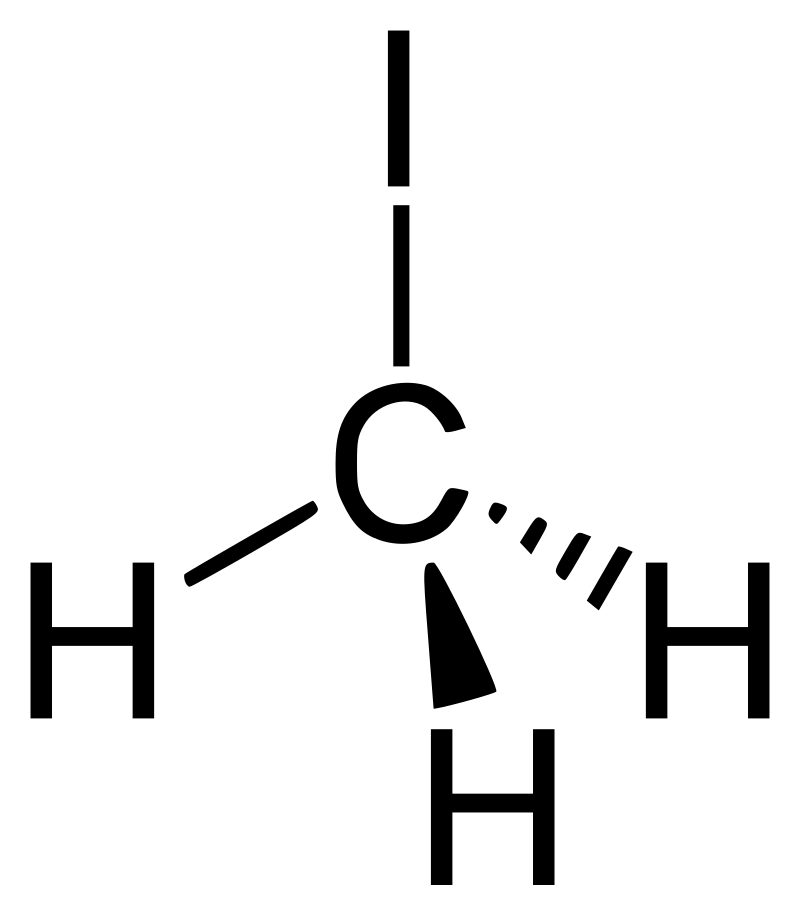
Iodomethane is a colorless, viscous liquid with a distinct odor, easily vaporizing into iodomethane vapors that float in air. Iodomethane melting point is -66.4°C. Iodomethane boiling point is 42.5°C. Iodomethane density is 2.28 g/ml. Iodomethane molecular weight is 141.95. It is miscible with common organic solvents such as ethanol and ether, while sparingly soluble in water. Exposure to sunlight causes it to decompose into elemental iodine, imparting a purple color. Hence, iodomethane is typically stored in sealed brown bottles, kept in cool, shaded areas, or sometimes with silver or copper wire inside the bottle to maintain stability.
In 1983, the production of iodomethane in the United States was approximately 50 tons (IARC, 1986). Due to its high refractive index, iodomethane is utilized for microscopic examination. It serves as an embedding material for diatoms, pyridine detection, a methylating agent in pharmaceuticals such as quaternary compounds, and in chemical synthesis. Additionally, it finds application as a photosensitive etchant in electronic circuits and as a component in fire extinguishers.
Iodomethane is a hazardous compound known for its volatility. Improper storage can lead to significant volatilization into the atmosphere, necessitating careful monitoring of iodomethane concentrations in the environment to prevent incidents of poisoning. In China, the maximum allowable concentration of hazardous substances in workplaces is set at 1 mg/m³. The determination of iodomethane concentration in the environment should adhere to GB/T16123-1995, using spectrophotometric methods with 1,2-naphthoquinone-4-sulfonate sodium.
Iodomethane (Iodomethane), also known as methyl iodide, is commonly abbreviated as "MeI."
Iodomethane is produced through an exothermic reaction by adding iodine to a mixture of methanol and red phosphorus, forming triiodophosphine in situ. Alternatively, it can be prepared by reacting methyl iodide with potassium iodide in the presence of calcium carbonate. Iodomethane can also be synthesized by reacting methanol with hydrogen iodide in aqueous solution.
Iodomethane is a hazardous compound. Acute inhalation exposure can lead to nausea, vomiting, dizziness, ataxia, slurred speech, drowsiness, blistering of the skin, and eye irritation in humans. Chronic (long-term) inhalation may affect the central nervous system and cause skin burns.
In summary, iodomethane, as an iodine-containing organic compound, has diverse chemical and industrial applications. Despite its importance in various fields, iodomethane poses potential environmental and health risks. Further research and regulation are necessary to balance its economic benefits with social responsibility, ensuring safe use and management.
[1] https://en.wikipedia.org/wiki/
[2] https://pubchem.ncbi.nlm.nih.gov/
[3] https://www.epa.gov/sites/default/files/
[4] https://www.ncbi.nlm.nih.gov/books/NBK498987/
[5] Ma, X. M. Study on hepatotoxicity of iodomethane in rats [Dissertation]. Jilin University, 2014.
 |
 |
 |