-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

Diethyl squarate is an important organic compound with wide-ranging applications and potential research value. As an ester compound, diethyl squarate plays a significant role in various fields such as chemical engineering and pharmaceuticals. Its synthesis methods, physicochemical properties, and applications in different domains have garnered attention from researchers. This article aims to systematically introduce the relevant knowledge of diethyl squarate, exploring its synthesis methods, characteristic properties, and potential application prospects, to provide reference and insight for research and development in related fields.
Diethyl squarate, a derivative of squaric acid, is a compound with the molecular formula C8H10O4, gaining increasing attention due to its widespread applications in scientific fields. Classified as an ester, diethyl squarate possesses a unique tetrahydroxy structure connecting two ester groups, conferring it with high reactivity. This reactivity enables it to readily participate in various chemical reactions, supporting its significance in materials science, drug development, and organic synthesis. Diethyl squarate has been used as an intermediate in the synthesis of perzinfotel. Due to its versatility, diethyl squarate proves to be a valuable tool for scientists, potentially unlocking advancements in many fields.
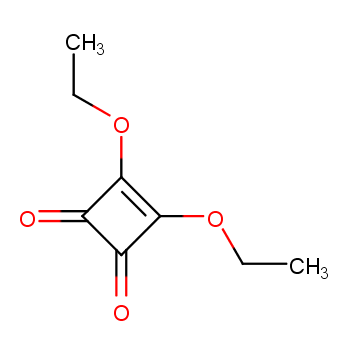
What is the density of diethyl squarate? The density of diethyl squarate at 25°C is approximately 1.15 g/mL. This relatively high density indicates that diethyl squarate is heavier than water per unit volume. Other key physical properties include a boiling point exceeding 95°C and a flash point >230°F.
Diethyl squarate exhibits significant reactivity, making it a valuable tool in various domains. Its ability for coupling reactions allows for the efficient formation of new carbon-carbon bonds. This feature finds applications in drug development, facilitating the synthesis of complex molecules with therapeutic potential. Acting as a coupling agent, diethyl squarate facilitates the formation of chemical bonds between amino groups on sugars and proteins. The products of these coupling reactions are termed neoglycoproteins, essentially glycoprotein carriers modified with oligosaccharides, serving as valuable tools in studying carbohydrate-protein interactions within biological systems.
In 150 mL of anhydrous ethanol, 3,4-dihydroxy-3-cyclobutene-1,2-dione (3.00 g, 26.4 mmol) was mixed with triethyl orthoformate (72.2 mmol, 12.0 mL) and stirred at room temperature. The mixture was then heated to 80°C and refluxed for 48 hours, followed by vacuum concentration. Purification of the crude yellowish oil was achieved by flash chromatography (DCM), yielding diethyl squarate as a yellowish oily substance (3.57 g, 97%).
In drug development and medicinal chemistry, diethyl squarate serves as a valuable tool. Its unique chemical reactivity allows for the effective synthesis of complex molecules with diverse functionalities. This means faster development of new drugs and improved versions of existing ones. Essentially a chemical building block, diethyl squarate simplifies the process of constructing complex molecules crucial for drug research.
Diethyl squarate plays a crucial role in materials science, particularly in crosslinking and polymerization reactions. These processes are vital for manufacturing robust and durable polymers with tailored properties for specific applications. By incorporating diethyl squarate, scientists can design polymers for various fields including coatings, adhesives, and composite materials. For example, the use of diethyl squarate in polymerization processes can lead to the development of tougher, more resilient coatings for automobiles or buildings.
Simone Stucchi et al. reported the use of diethyl squarate as a crosslinking agent in gelatin hydrogels. Here's the role of diethyl squarate in this scenario:
Essentially, diethyl squarate plays a crucial role in manufacturing stronger, more functional gelatin hydrogels, opening doors for its applications in the biomedical field.
Diethyl squarate, once considered a niche compound, is increasingly gaining attention across scientific fields due to its unique properties. Here are some exciting trends in its usage:
While valuable, diethyl squarate requires careful handling due to its inherent nature. Standard safety protocols for laboratory chemicals should be strictly followed. Always wear appropriate personal protective equipment (PPE) such as gloves, safety goggles, and chemical-resistant lab coats when handling diethyl squarate. Minimize inhalation risks by using a fume hood and maintain a tidy workspace. It is crucial to stay informed about the latest legal requirements and regulations for handling and disposal of diethyl squarate set by governing bodies. Referring to safety data sheets (SDS) for specific procedures and proper waste disposal methods is essential for ensuring a safe and compliant working environment.
In conclusion, diethyl squarate is an important organic compound with broad prospects in chemical engineering, pharmaceuticals, and other fields. Through this article's introduction, we have explored the synthesis methods, characteristic properties, and novel applications of diethyl squarate. With the continuous advancement of science and the growing demand for green, environmentally friendly products, the application of diethyl squarate is expected to be further optimized and expanded. It is hoped that this article will promote a deeper understanding of diethyl squarate, drive research and development in related fields, and lay a solid foundation for future applications and innovations.
[1] https://link.springer.com/content/pdf/10.1007/BF00731506.pdf
[2] Ryan P, Shi Y, von Itzstein M, et al. Novel bisubstrate uridine-peptide analogues bearing a pyrophosphate bioisostere as inhibitors of human O-GlcNAc transferase[J]. Bioorganic Chemistry, 2021, 110: 104738.
[3] https://pubmed.ncbi.nlm.nih.gov/8737256/
[4] https://pubs.acs.org/doi/10.1021/acs.langmuir.1c02080
[5] https://en.wikipedia.org/wiki/Squaric_acid
 |