Diphenylsilanediol is a significant organosilicon compound with unique structure and properties, finding wide applications in organic synthesis and materials science. This article will explore the properties of diphenylsilanediol and its applications in organic synthesis and materials science, aiming to provide readers with a comprehensive understanding of this important organosilicon compound.
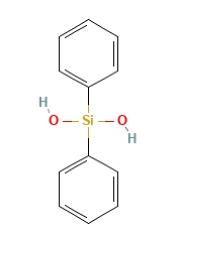
Diphenylsilanediol, Ph2Si(OH)2, is a silanol compound. Tetrahedral molecules form hydrogen-bonded columns in the solid state, and it can be prepared by hydrolysis of diphenyldichlorosilane Ph2SiCl2. Diphenylsilanediol can act as an anticonvulsant, similar to phenytoin. Although the compound is stable under normal conditions, the presence of basic impurities can accelerate the condensation of silanol groups. The structure of diphenylsilanediol is shown below:

Solubility refers to the maximum amount of a substance (solute) that can dissolve in a specific solvent to form a stable solution under certain temperature and pressure conditions. When no more solute can dissolve, the solution is considered saturated.
(1) Similarity between Solute and Solvent: Polar solutes dissolve better in polar solvents, while nonpolar solutes dissolve better in nonpolar solvents. This is because intermolecular forces (such as dipole-dipole interactions and hydrogen bonding) between solute and solvent molecules influence their attraction and mixing ability.
(2) Functional Groups: The presence of functional groups in solute molecules can significantly affect their solubility. For example, hydroxyl groups (-OH) can form hydrogen bonds with water, making the solute more soluble in water.
(3) Molecular Size and Shape: Generally, smaller and more compact molecules have better solubility than larger and more complex molecules.
(4) Temperature: In most cases, solubility increases with temperature. With increasing temperature, solvent molecules gain kinetic energy, allowing them to disrupt solute-solute interactions and promote solvation.
Diphenylsilanediol is soluble in acetone, ether, ethyl alcohol, and dimethylformamide. It is almost insoluble in water, xylene, toluene, n-hexane, benzene, and carbon tetrachloride.
Diphenylsilanediol finds utility in the pharmaceutical field, electronics industry, and polymer materials. What is Diphenylsilanediol used for? Diphenylsilanediol has several key applications:
Diphenylsilanediol serves as a fundamental raw material for producing phenylsiloxane fluids. It is also used as a structural control agent for silicone rubber and intermediate for other organosilicon compounds.
Diphenylsilanediol is employed in the production of organic polymers with high refractive indices and high transmittance. These polymers are primarily used in LED or electronic encapsulation materials.
Diphenylsilanediol can be used to synthesize linear ethylene-based oligosiloxane resins, which can potentially be used for manufacturing thermally resistant light-emitting diode (LED) encapsulants. Bae et al. studied the color stability of high refractive index phenyl-siloxane hybrid materials in LED encapsulation. Phenyl hybrids were prepared by the hydrogenation reaction of linear ethylene-based oligosiloxanes and hydrogen-based oligosiloxane resins using a platinum (Pt) catalyst. Linear oligosiloxanes were synthesized through a non-hydrolytic sol-gel condensation reaction of silicon precursors with bifunctional groups containing vinyl, hydrogen, and phenyl. Compared to branched oligosiloxanes, linear oligosiloxanes exhibited higher condensation degrees and larger molecular sizes, resulting in lower shrinkage rates and effective curing properties. Phenyl hybrids displayed high optical transparency with a refractive index of up to 1.57. Particularly, minimal usage of Pt catalyst was achieved at 180 °C to suppress yellowing of hybrids during prolonged aging. These results suggest that phenyl hybrids derived from linear oligosiloxanes can serve as high-performance LED encapsulants.
Utilizing diphenylsilanediol, thiophene-derived dielectric layers of organic thin-film transistors can be synthesized.
Studies indicate that diphenylsilanediol may serve as an anticonvulsant drug similar to phenytoin. However, further development may be required before it becomes a therapeutic agent.
Reported by J. Keith Fawcett et al., the crystal and molecular structures of diphenylsilanediol (a compound most proven to have anticonvulsant activity) have been determined and compared its stereochemistry with other antiepileptic drugs. The crystals of diphenylsilanediol belong to the triclinic system, space group, cell dimensions a = 9.912, b = 15.048, c = 14.519 Å, α = 120.83, β = 99.95, γ = 100.84°, Z = 6 molecules per cell (3 molecules per asymmetric unit). The structure was determined by Patterson and Fourier methods and refined to final difference value R = 0.034. The phenyl ring planes in the molecule rotate 80° relative to each other, with a distance of 2.66 Å between hydroxyl oxygen atoms. Molecular parameters help describe the limits of stereochemical changes in these molecules and retain anticonvulsant activity.
Diphenylsilanediol is a compound that may pose dangers if mishandled. Safety Data Sheets (SDS) provide crucial information about material hazards and how to handle and store it safely.
According to the 2012 OSHA Hazard Communication Standard (29 CFR 1910.1200), this chemical is considered hazardous.
(1) Thoroughly wash face, hands, and any exposed skin after handling.
(2) Wear protective gloves/clothing/eye protection/face protection.
(3) Avoid inhaling dust/fume/gas/mist/vapors/spray.
(4) Use only outdoors or in a well-ventilated area.
(5) Keep away from heat/sparks/open flames/hot surfaces. - No smoking.
(6) Ground/bond container and receiving equipment.
(7) Use explosion-proof electrical/ventilation/lighting equipment.
If symptoms persist, seek medical advice.
Immediately flush eyes with plenty of water, also under the eyelids, for at least 15 minutes. Get medical attention.
Immediately flush skin with plenty of water for at least 15 minutes. If skin irritation persists, seek medical attention.
Move to fresh air. If breathing is difficult, give artificial respiration. If symptoms occur, seek medical attention.
Rinse mouth with water, then drink plenty of water. If symptoms occur, seek medical attention.
Wear personal protective equipment/face protection. Ensure adequate ventilation. Do not get in eyes, on skin, or on clothing. Avoid ingestion and inhalation. Prevent dust formation.
Keep container tightly closed in a dry, cool, well-ventilated place. Keep away from incompatible materials, water, oxidizing agents.
In conclusion, diphenylsilanediol is an organosilicon compound of significant utility. Its unique structure and properties render it widely applicable in organic synthesis and materials science. Through the discussion in this article, we have gained insights into the nature and applications of diphenylsilanediol, providing essential references and insights for research and practice in relevant fields.
[1] Bae J, Kim Y H, Kim H Y, et al. Sol–gel synthesized linear oligosiloxane-based hybrid material for a thermally-resistant light emitting diode (LED) encapsulant[J]. RSC advances, 2013, 3(23): 8871-8877.
[2] https://cdnsciencepub.com/doi/10.1139/v77-510
[3] https://www.fishersci.com
[4] https://pubs.acs.org/doi/pdf/10.1021/ac60157a047
[5] https://en.wikipedia.org/wiki/Diphenylsilanediol
 |
 |