Fenoxaprop-p-ethyl herbicide is a commonly used herbicide, widely applied in the agricultural field. It can effectively control certain grass weeds, including some that are resistant to other herbicides. Fenoxaprop-p-ethyl herbicide has a fast and persistent weed-killing effect, with high crop safety, thus favored by farmers and agricultural experts. In modern agricultural production, fenoxaprop-p-ethyl herbicide plays an important role, providing an effective weed control method, helping farmers protect crops, and improve yield and quality. Therefore, understanding the uses and characteristics of fenoxaprop-p-ethyl herbicide is of significant importance for agricultural production.
Fenoxaprop-p-ethyl herbicide, also known as Fenoxaprop-P-ethyl, is a synthetic compound not naturally occurring. It is the R-isomer of fenoxaprop, which exists as R and S forms. Fenoxaprop-P-ethyl functions as a post-emergence herbicide, controlling annual and perennial grass weeds in crops such as wheat, barley, and established perennial broadleaf crops. The structural formula of Fenoxaprop-P-ethyl is as follows:
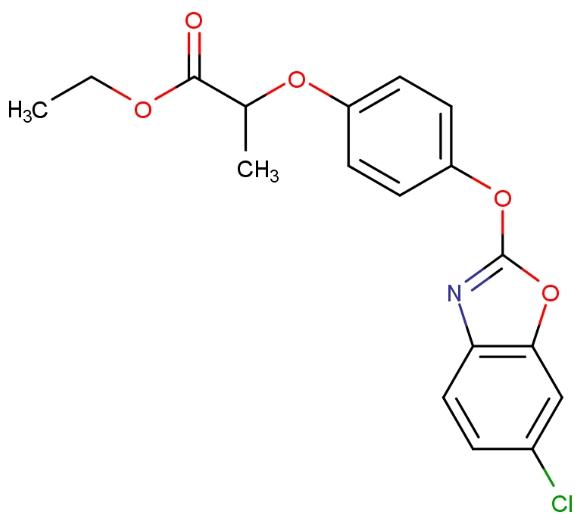
Fenoxaprop-p-ethyl herbicide is an acetyl-coenzyme A carboxylase (ACCase) inhibitor developed by Bayer Crop Science, belonging to the aryloxyphenoxy propionate selective internal absorption herbicide class. The herbicide inhibits the carboxylation of acetyl-coenzyme A, thereby disrupting the biosynthesis of fatty acids and compromising the integrity of the cell membrane, inhibiting the growth of plant meristems and disrupting normal plant metabolism, ultimately leading to the death of sensitive plants. The use of fenoxaprop-p-ethyl herbicide includes controlling grass weeds in broadleaf crops such as rapeseed, soybean, cotton, and peanuts. When combined with the safener cloquintocet-mexyl, it enhances wheat's resistance to fenoxaprop-p-ethyl for controlling annual and perennial grass weeds.
Fenoxaprop-p-ethyl herbicide belongs to the aryloxyphenoxy propionate (AOPP) herbicide class, also known as FOPs, which mimic natural plant hormones.
Chemical formula: C18H16ClNO5
CAS number: 71283-80-2
Molar mass: 361.78 g/mol
Appearance: Colorless to pale yellow liquid
Physical state: Liquid
Boiling point: >300 ℃
Melting point: 79-84℃ (lit.)
Density: 1.31 g/cm3
Refractive index: 1.62
Fenoxaprop-p-ethyl is the active ingredient in several herbicide products. It is usually formulated as an emulsifiable concentrate (EC) or soluble concentrate (SC) to be diluted with water before application. Commercial products may also contain inactive ingredients such as surfactants, solvents, and defoamers, which aid in formulation, mixing, and application of the herbicide.
What is Fenoxaprop-p-ethyl used for? Fenoxaprop-p-ethyl herbicide is a post-emergence herbicide used to control annual and perennial grasses, proven effective through field trials and extensive global usage. It is typically supplied as an oil emulsion, mixed with water and applied as a spray.
Examples of weed control include: Crabgrass; Goosegrass; Bermuda grass; Foxtails; Barnyard grass. Application examples include: Turfgrass; Ornamental plants; Rice; Cereals including wheat, barley, rye, and oats.
As a highly efficient herbicide, fenoxaprop-p-ethyl herbicide has been widely used in wheat fields to control grass weeds such as wild oats. However, both long-term and one-time use of fenoxaprop-p-ethyl herbicide in wheat fields has led to resistance in wild oats. Compared to other herbicides, fenoxaprop-p-ethyl herbicide acts only on ACCase, making weeds more likely to develop resistance. Fenoxaprop-p-ethyl herbicide is classified as a high-risk level for resistance, with weeds typically developing resistance after one year of continuous use. Recently, controlling wild oats in wheat fields solely with fenoxaprop-p-ethyl has become difficult.
Fenoxaprop-P-ethyl is a post-emergent herbicide used to control both annual and perennial grasses. It exhibits low water solubility and volatility. While fenoxaprop-p-ethyl herbicide typically does not persist in soil, under certain conditions, it may persist in aquatic systems. It poses low acute oral toxicity to birds and bees; moderate toxicity to mammals, earthworms, fish, and aquatic invertebrates. Its oral toxicity to humans is considered moderate to high, and it is recognized as an irritant.
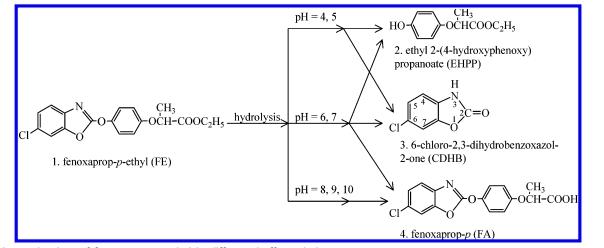
Fenoxaprop-P-ethyl poses a risk to aquatic environments, necessitating avoidance of direct contamination of aquatic habitats (Asshauer et al., 1990). Due to the hydrolysis of Fenoxaprop-P-ethyl, Fenoxaprop acid (FA) has been observed in plants, microorganisms, and the natural environment (Wink & Luley, 1988). Increased usage of parent herbicide Fenoxaprop-P-ethyl may pose a risk to groundwater resources due to the fluidity and acidity of FA.
Fenoxaprop-P-ethyl is widely used in agriculture as a post-emergence aryloxyphenoxy propionate (AOPP) herbicide.
Microbial metabolism is the primary mechanism leading to the degradation of Fenoxaprop-P-ethyl in natural soils, without harmful metabolites (Lin et al., 2008). To date, there have been no reports of pure culture microorganisms capable of complete degradation of Fenoxaprop-P-ethyl and other AOPP herbicides. Mixed microbial populations can utilize Fenoxaprop-P-ethyl (15 mg L?1) as their sole carbon and nitrogen source, with additional carbon sources increasing the metabolic rate, resulting in FA and 6-chloro-2,3-dihydrobenzothiazole-2-one (CDHB) as metabolites (Gennari et al., 1995).
Weiliang Dong et al. isolated a highly efficient Fenoxaprop-P-ethyl-degrading strain DL-2 from enrichment cultures, identified as an Acinetobacter sp. Through HPLC/MS analysis, metabolite fenoxaprop acid (FA) was identified. Strain DL-2 can also degrade various other AOPP herbicides. A new FE hydrolytic esterase gene afeH was cloned from strain DL-2 and functionally expressed in Escherichia coli BL21(DE3). The specific activity of recombinant AfeH towards Fenoxaprop-P-ethyl was 216.39 U mg(-1), with Km and Vmax values of 0.82 μM and 7.94 μmol min(-1) mg(-1), respectively. AfeH can also hydrolyze various AOPP herbicides, nitrobenzene esters, and triglycerides. The optimal pH and temperature for AfeH were 9.0 and 50°C, respectively; the enzyme was activated by Co(2+) and inhibited by Ca(2+), Zn(2+), and Ba(2+). AfeH was strongly inhibited by benzylsulfonyl and SDS, with weaker inhibition by dimethyl sulfoxide.
Fenoxaprop-P-ethyl serves as an irreplaceable herbicide in agriculture, offering efficient and broad-spectrum weed control. Its rapid and effective weed control helps mitigate interference from grassy weeds on crops, thereby aiding farmers in enhancing crop yield and quality. However, when using Fenoxaprop-P-ethyl herbicide, adherence to usage instructions is imperative to ensure its safe and effective application and to mitigate potential impacts on the environment and human health. By understanding the uses and characteristics of Fenoxaprop-P-ethyl herbicide, we can better utilize this herbicide to achieve efficient and sustainable agricultural production, delivering improved economic benefits and agricultural experiences for farmers.
[1] Zhu, W., Yan, D., Li, L., et al. (2019). Effects of Fenoxaprop-P-ethyl on controlling gramineous weeds in rapeseed fields and its influence on light and nutrients. Journal of Jiangxi Agricultural University, 31(04), 56-60. DOI:10.19386/j.cnki.jxnyxb.2019.04.11.
[2] https://www.biorxiv.org/content/10.1101/549402v1.full
[3] https://sitem.herts.ac.uk/aeru/ppdb/en/Reports/303.htm
[4] https://pubmed.ncbi.nlm.nih.gov/25812814/
[5] https://www.sciencedirect.com/science/article/abs/pii/S0960852415003697
[6] https://pubchem.ncbi.nlm.nih.gov/compound/91707
 |
 |
 |