-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

In the realm of drug synthesis and application, research on the synthesis methods and applications of compounds like Flavopiridol, which hold potential therapeutic uses, garners considerable attention. This article explores the synthesis of Flavopiridol and its medical applications, aiming to provide readers with deeper insights into the compound's synthesis process and potential value.
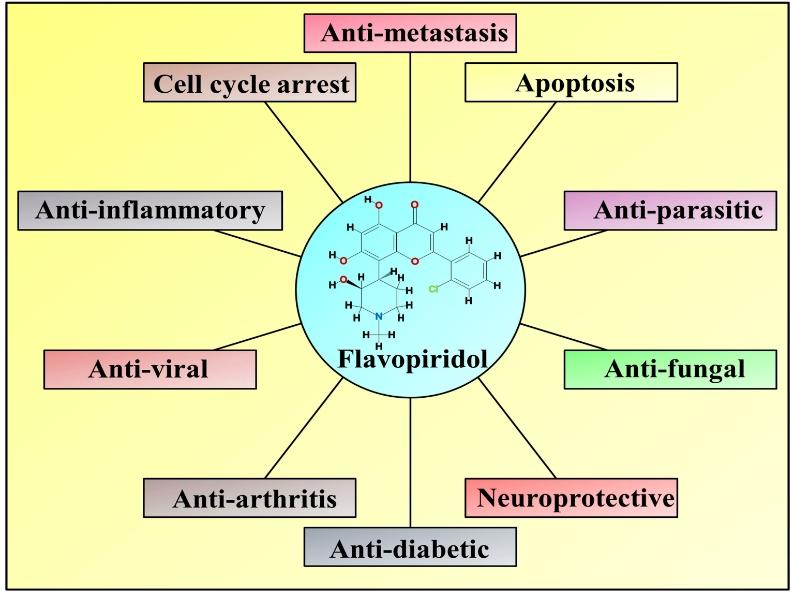
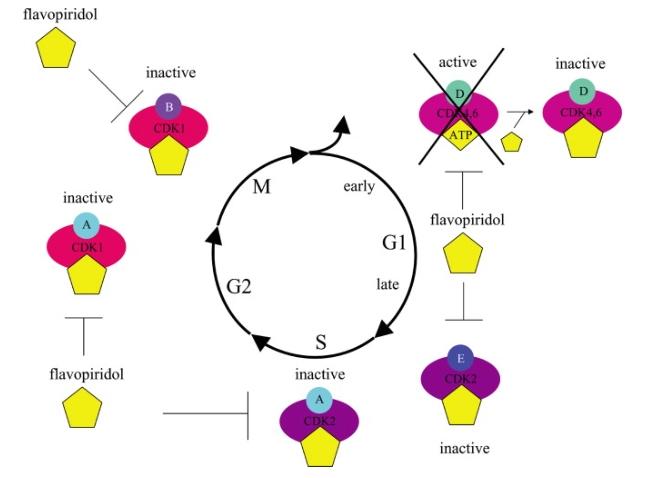
Flavopiridol, a flavone synthesized from the natural product rohitukine, derived from the medicinal plant Dysoxylum binectariferum Hiern in India. A deeper understanding of the biological mechanisms of action of these molecules could lead to the development of effective therapeutic strategies against various life-threatening diseases such as cancer, viruses, fungal infections, parasites, and neurodegenerative diseases. Flavopiridol's mechanism of action reveals its potential to inhibit CDKs (cyclin-dependent kinases) and other kinases, thereby suppressing various processes including cell cycle progression, apoptosis, tumor proliferation, angiogenesis, tumor metastasis, and inflammatory processes. Synthesized derivatives of flavopiridol overcome some drawbacks of the parent compound, significantly enhancing CDK inhibition activity and the ability to treat severe human diseases. Flavopiridol appears poised to become a candidate drug for formulating comprehensive strategies against and alleviating human diseases.
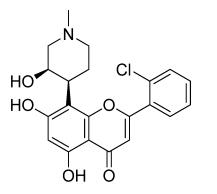
Flavopiridol's chemical name is 2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydroxy-1-methyl-4-piperidinyl]benzo[b]pyran-4-one. The chemical structure of flavopiridol is depicted in the figure below.
The crystal structure of flavopiridol in complex with CDK2 has been determined using X-ray crystallography, revealing the molecular basis for its inhibition of CDKs. The crystallographically determined molecular structure describes the binding reaction of the drug at the CDK2 active site. The chlorine atom of flavopiridol interacts favorably with the active site of CDK2, which may contribute to flavopiridol's increased kinase inhibition. On April 23, 2014, the U.S. Food and Drug Administration (FDA) granted orphan drug status to flavopiridol for the treatment of AML patients. On November 17, 2014, The Tolero Pharmaceuticals, Inc. announced flavopiridol's selection as one of the top ten most promising oncology projects.
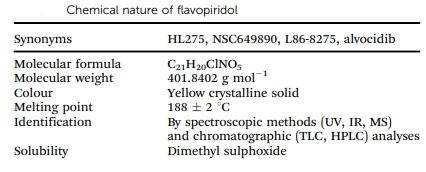
Flavopiridol is also known as alvocidib, L86-8275, HL275, and NSC649890. This flavone compound is synthesized from natural anti-rheumatic flavonoids. Flavopiridol is a yellow crystalline solid, identified through various spectroscopic and chromatographic techniques. Its molecular formula is C21H20ClNO5; its melting point ranges from 186°C to 190°C, and its molecular weight is 401.84 g mol−1. Flavopiridol exhibits minimal solubility in water; however, it is soluble in organic solvents including ethanol, dimethyl sulfoxide, and dimethylformamide. The table below lists the chemical properties of flavopiridol, including its molecular formula, color, melting point, and solubility.
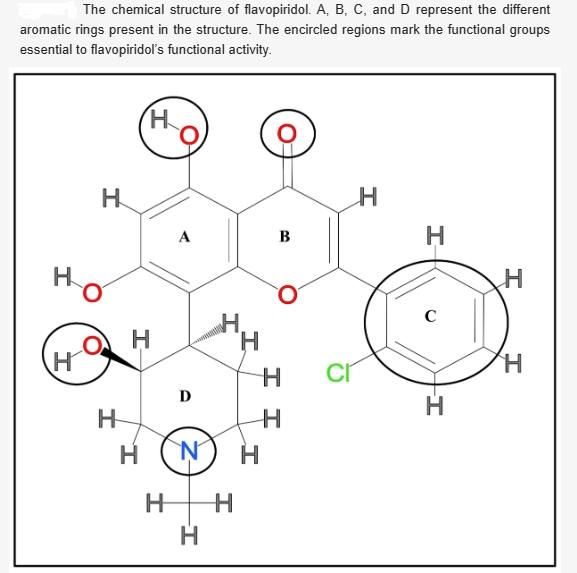
The crystal structure of flavopiridol has been determined in complex with cyclin-dependent kinase 2 (CDK2), where the flavone nucleus of the former binds to the ATP binding pocket of the latter. It exhibits CDK inhibition activity via the inclusion of the d-ring (also known as the 3-hydroxy-1-methylpiperidinyl ring), as depicted in the figure below. In contrast, this ring is absent in two structurally related natural flavonoids, quercetin and fisetin, which exhibit poor CDK inhibition activity.
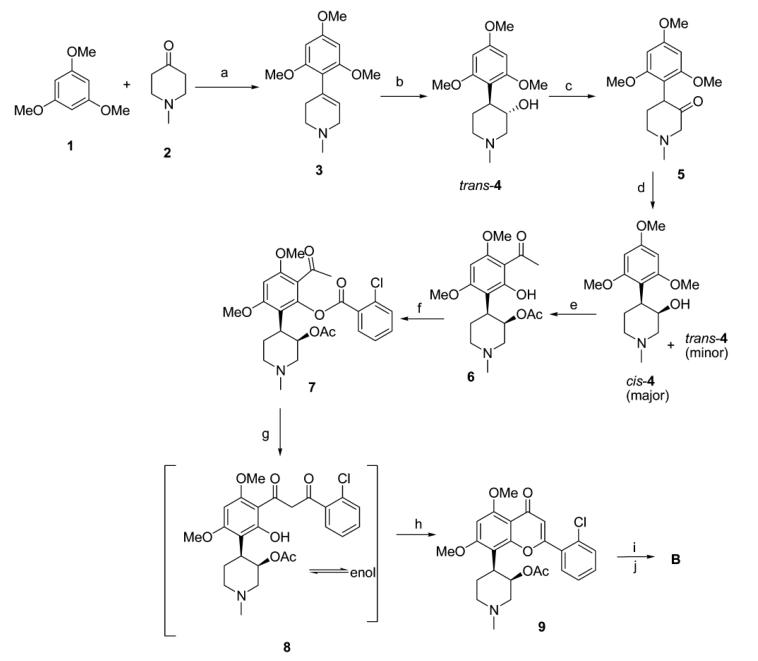
In the synthesis of flavopiridol, as depicted in the figure below, the first step involves the condensation of 1,3,5-trimethoxybenzene 1 with 1-methyl-4-piperidone 2 in the presence of hydrogen chloride-saturated acetic acid to generate the alkene 3, which is converted to the trans-aryl piperidine alcohol trans-4 through hydroboration with borane. The cis-aryl piperidine alcohol cis-4 is obtained via stereochemical inversion of alcohol through two steps: oxidation of cis-4 to the corresponding ketone 5, followed by reduction using sodium borohydride. The resulting product is a mixture of cis/trans alcohols (cis-4 and trans-4 in a 7:3 ratio determined by GC analysis).
The separation of cis/trans alcohols is achieved through fractional crystallization of their enantiomer salts with (-)-dibenzoyl-D-tartaric acid or separation of their enantiomeric ester mixtures with (-)-methyl oxirane (silica gel fast chromatography). In the presence of trifluoroethoxyborane, acetylation of cis-4 is carried out to obtain 6 using acetic anhydride. Benzoylation of 6 with 2-chlorobenzoyl chloride yields the benzoate 7. The formation of flavone involves a neighboring substitution between the benzoate functional group and benzophenone. Treatment of 7 with KOH in pyridine reflux affords the rearranged intermediate 8, which upon dehydration with acetic acid and sulfuric acid yields 9. Deacetylation followed by demethylation yields flavopiridol B.
Flavopiridol presents a promising anticancer drug, yet its clinical application faces obstacles due to limitations in production. Optimizing the synthesis process is crucial for making it a more viable treatment option. A key strategy involves streamlining current synthetic steps to enhance efficiency and reduce waste. This may entail exploring alternative reagents or catalysts that improve reaction rates and product yields.
However, achieving high yield and purity remains a significant challenge. Flavopiridol is a complex molecule prone to producing undesirable by-products during synthesis. Developing efficient, scalable purification methods that maintain high yields and purity without sacrificing overall yield is essential to making flavopiridol more readily accessible and economically viable for therapy.
Flavopiridol, a synthetically derived compound, holds therapeutic potential for treating various cancers. It can inhibit cyclin-dependent kinases essential for cell division, thereby disrupting the uncontrolled growth of cancer cells. By synthesizing flavopiridol in laboratory settings, researchers obtain a reliable source of this crucial compound for further study. This paves the way for deeper exploration of its effectiveness in different cancer treatments and potential clinical trials.
The ability to synthesize complex molecules like flavopiridol demonstrates the power of synthetic technology. It grants scientists the freedom to design and produce specific compounds for research or other purposes. This not only drives medical advancements but also has far-reaching implications across industries. Synthetic technology can create new drugs, ultimately shaping the process of scientific discovery and medical innovation.
Through the discussion of Flavopiridol synthesis and application in this article, readers gain a comprehensive understanding of the characteristics and potential value of this compound. Flavopiridol, as a promising drug, holds broad prospects in fields such as cancer treatment. With continuous advancements in scientific technology and deeper research, the synthesis methods and applications of Flavopiridol are expected to undergo further exploration and development, providing more valuable references for drug research and clinical treatment.
[1] Deep A, Marwaha R K, Marwaha M G, et al. Flavopiridol as cyclin dependent kinase (CDK) inhibitor: a review[J]. New Journal of Chemistry, 2018, 42(23): 18500-18507.
[2] Joshi H, Tuli H S, Ranjan A, et al. The Pharmacological Implications of Flavopiridol: An Updated Overview[J]. Molecules, 2023, 28(22): 7530.
 |
 |
 |