What is another name for isovaleraldehyde? Isovaleraldehyde, also known as 3-methylbutanal, with isovaleraldehyde CAS NO. 590-86-3, is a versatile chemical product used to create a range of high-value-added products. It is designated as an edible flavoring in China, contributing to various fruit-flavored essences. Additionally, its derivatives such as isovaleric acid, isovaleramide, and esters are utilized in edible and personal care fragrances.
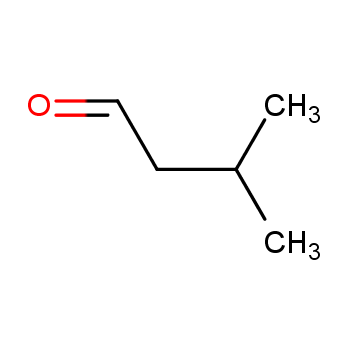
Isovaleraldehyde is a colorless liquid at room temperature with several physical properties that are critical to its industrial applications and safety precautions. What is the flash point of isovaleraldehyde? Here are some of the key properties:
(1) Isovaleraldehyde Color: Clear colorless to pale yellow
(2) Isovaleraldehyde Smell: Present in natural oils like orange, lemon, peppermint, and eucalyptus. Also found in processed foods such as beer, cheese, and chocolate, it exhibits a variety of descriptors from "apple-like" to "suffocating."
(3) Isovaleraldehyde Melting Point: -51°C (-60°F; 222K)
(4) Isovaleraldehyde Boiling Point: 92°C (198°F; 365K)
(5) Isovaleraldehyde Density: 0.785 g/mL at 20°C (lower than water)
(6) Isovaleraldehyde Vapor Density: 2.96 (air = 1)
(7) Isovaleraldehyde Vapor Pressure: 50 mmHg at 30°C
(8) Isovaleraldehyde Solubility: Soluble in alcohols and ethers, slightly soluble in water
(9) Isovaleraldehyde Flash Point: 29°F
(10) Isovaleraldehyde Refractive Index n20/D 1.388 (lit.)
Isovaleraldehyde features a five-carbon chain structure with the molecular formula (CH3)2CHCH2CHO. It includes a methyl group ((CH3)2) attached to the third carbon and terminates with an aldehyde group (CHO) at the chain end. This structural arrangement exhibits unique characteristics:
(1) Chirality: The presence of the third carbon bonded to four different groups (two methyl groups, one hydrogen, and the remaining chain) makes isovaleraldehyde a chiral molecule, existing in two non-superimposable mirror image forms.
(2) Polarity: The carbonyl group introduces polarity within the molecule, resulting in low solubility in water.
By examining these aspects, the distinct properties and applications of isovaleraldehyde in various industries are underscored, highlighting its role in fragrance creation and beyond.
Isoamyl alcohol, abundant and low-cost, is widely present as a byproduct in alcohol fermentation at distilleries and can be obtained by distillation. Therefore, the oxidative dehydrogenation of isoamyl alcohol to produce isovaleraldehyde is considered a relatively economical process.

(1) According to the "Chinese Chemical Commodity Encyclopedia," sulfuric acid and chromic acid can directly oxidize isoamyl alcohol to isovaleraldehyde. Researchers at Guangxi University, led by Li Jinglin, utilized Ag/KZSM-5 molecular sieves as catalysts to react isoamyl alcohol with air in specific proportions to obtain isovaleraldehyde. Additionally, Gao Hufei and colleagues developed a novel catalyst, Co-doped ZnO/MCM-41 molecular sieves, for the catalytic oxidation of isoamyl alcohol, although these methods are complex to operate, pose significant safety hazards, and suffer from low yields, making large-scale industrial production impractical.
(2) Recep Ozen developed a stable, easily storable, and low-cost chromium reagent, 3,4-dihydroisoquinoline trichloroacetic acid chromium, which, under non-protonic solvent and room temperature conditions, can effectively oxidize primary and secondary alcohols to carbonyl compounds, including aryl compounds. Using this oxidant, oxidation of isoamyl alcohol yielded isovaleraldehyde in just half an hour with a 90% yield.
Despite the maturation of processes for synthesizing isovaleraldehyde from isoamyl alcohol, the starting material, isoamyl alcohol, often has low purity and contains impurities, necessitating purification before participation in reactions, thereby increasing production costs. Therefore, the hydroformylation of isobutene to produce isovaleraldehyde is more commonly employed in industry. This reaction boasts atom economy, achieves zero emissions of waste, is highly efficient, energy-saving, and relatively safe.

Given that isovaleraldehyde can be extracted from leucine, its presence is not limited to beer. This compound is found as a flavoring component in various foods, described as having a malty flavor, and is present in cheese, coffee, chicken, fish, chocolate, olive oil, and tea.
The compound is used as a reactant in synthesizing various compounds. Notably, it is employed in synthesizing 2,3-dimethylbut-2-ene, which is then converted to 2,3-dimethylbutane-2,3-diol and methyl tert-butyl ketone, commonly known as mesotrione. Mesotrione itself is used in the synthesis of numerous herbicides. Additionally, a range of drugs, such as butethamine, are synthesized from isovaleraldehyde and its corresponding acids. It is a common reagent or building block in organic synthesis.
Isovaleraldehyde undergoes cyclotrimerization under acidic conditions to yield 2,4,6-triisobutyl-1,3,5-trioxane. This is a flavoring agent used in candies, tobacco, and other foods, toothpaste, etc. It is claimed to impart creamy, dairy, vanilla-chocolate, and berry flavors.
According to IFF, isovaleraldehyde is used as a food flavoring additive.
Isovaleraldehyde, as an important organic compound, finds widespread application in the flavor and fragrance industry due to its distinctive aromatic properties. Its stability and excellent aroma effects make it an indispensable component in many products. However, its potential irritant effects on the skin and respiratory system necessitate careful and safe management in industrial and consumer applications.
[1] Sun Yanan. Rhodium-Catalyzed Hydroformylation of Isobutene to Prepare Isovaleraldehyde. Shanghai Normal University, 2018.
[2] https://en.wikipedia.org/wiki/Isovaleraldehyde
[3] https://pubchem.ncbi.nlm.nih.gov/
[4] https://www.acs.org/molecule-of-the-week/archive/i/isovaleraldehyde.html
 |
 |
 |