-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here


Lewis structures, devised by Gilbert N. Lewis, visually represent electron arrangements in molecules. By depicting valence electrons as dots and bonds as lines, Lewis structures predict a molecule's shape and properties based on the octet rule. This rule states that atoms tend to achieve stability by having eight electrons in their outer shell. Lewis structures adhere to this rule, offering a clear picture of chemical bonding.
Potassium bromide (KBr) is a white crystalline solid composed of potassium (K) and bromine (Br) atoms. It is commonly used in photography, as a flame retardant, and in various pharmaceutical applications. KBr is highly soluble in water and exhibits strong ionic bonding between the potassium and bromine atoms.
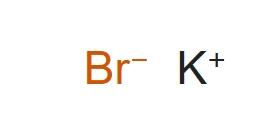
Let's dive into drawing the lewis dot structure for potassium bromider:
Step 1: Identify the Central Atom: Potassium (K) and Bromine (Br) are the atoms involved. Since KBr is an ionic compound, we represent them separately.
Step 2: Calculate Total Valence Electrons: Potassium contributes 1 valence electron, and bromine contributes 7, but since it's an ionic compound, we show K+ and Br- ions.

Step 3: Arrange Electrons Around Atoms: Represent potassium as K+, and bromine as Br-, with 8 valence electrons (lone pairs) around the bromine atom.
Step 4: Fulfill the Octet Rule: The bromine atom should have 8 electrons (2 lone pairs and 6 bonding pairs). Potassium will lose its valence electron to form a K+ ion.
Step 5: Check for Formal Charges: Formal charges are not necessary here since it's an ionic compound.
Since KBr is an ionic compound, it does not have a typical molecular geometry. Instead, it forms a crystal lattice structure where each potassium ion (K+) is surrounded by bromide ions (Br-) and vice versa. The arrangement is typically cubic or orthorhombic.
| Potassium Bromide Cas 7758-02-3 | |
| Molecular formula | KBr |
| Molecular shape | Ionic crystal lattice |
| Polarity | Nonpolar (ionic) |
To determine if a Lewis structure is polar, examine the molecular geometry and bond polarity. In the case of potassium bromide (KBr), the Lewis structure shows potassium (K+) and bromine (Br-) ions. KBr is an ionic compound, which means it is nonpolar overall due to the complete transfer of electrons and the resulting ionic bonding.
For ionic compounds like KBr, bond energy can be determined using the lattice energy, which is the energy required to separate the ions in the crystal lattice. Lattice energy for KBr is approximately 682 kJ/mol, representing the energy required to break the ionic bonds in one mole of KBr.
Bond order is the number of chemical bonds between a pair of atoms. In the Lewis structure of KBr, the bond order is 1, indicating a single ionic bond between potassium (K+) and bromine (Br-).
Electron groups in a Lewis structure include both bonding pairs (shared electrons) and lone pairs (non-bonded electrons) around an atom. In KBr, potassium (K+) is represented as a bare cation, and bromine (Br-) is surrounded by 8 valence electrons (lone pairs).
In a Lewis dot structure, the dots represent valence electrons. Each dot corresponds to one valence electron of an atom. In KBr, potassium is represented as K+, and bromine is represented by 8 dots (lone pairs) around the bromine atom. The dots help visualize how electrons are shared or paired between atoms.
When determining the best Lewis structure for KBr, it's important to consider both the bonding and the arrangement of electrons to ensure the most stable representation. Choosing the correct structure helps in understanding its molecular properties and behavior. If you're exploring how to choose the best Lewis structure for KBr or other compounds, Guidechem provides access to a wide range of global suppliers of Potassium bromide. Here, you can find the ideal raw materials to support your research and applications.
 |
 |