-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

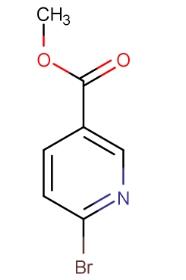
Methyl 6-bromonicotinate, with the CAS number 26218-78-0 and the molecular formula C7H6BrNO2, is a widely used chemical reagent in organic synthesis. It is particularly valuable in the synthesis of various heterocyclic compounds. Often used as a starting material for synthesizing nicotinic acid derivatives and pyridine-based ligands, it is also a potential hypolipidemic agent and nicotinic acid analog. It affects the mobilization of free fatty acids in the body. The structure of Methyl 6-bromonicotinate is as follows:
In Organic Synthesis
As a synthetic building block, Methyl 6-bromonicotinate can introduce various functional groups through electrophilic substitution reactions of the bromine atom, thereby constructing structurally diverse organic molecules. It plays a significant role in the synthesis of heterocyclic compounds, providing a rich molecular library for drug and pesticide development.
In Drug Development
In the field of medicinal chemistry, Methyl 6-bromonicotinate is used as a key precursor in the synthesis of potential hypolipidemic drugs. These drugs help regulate cholesterol levels, thus playing a role in treating cardiovascular diseases. Methyl 6-bromonicotinate is crucial in the preparation of nicotinic acid analogs known for their lipid-lowering effects.
Catalysis Research
Methyl 6-bromonicotinate also has significant applications in catalysis research, particularly in the development of catalysts for cross-coupling reactions. These reactions involve the bonding of carbon atoms, which is a key step in synthesizing various organic compounds.
In Agrochemicals
In the agricultural sector, Methyl 6-bromonicotinate can be used as a starting material for synthesizing novel pesticides or herbicides. Its potential impact on fatty acid metabolism makes it promising for developing new insecticides.
In Materials Science
The reactivity of Methyl 6-bromonicotinate also offers potential applications in materials science. By introducing this compound, surface properties of materials can be modified, or new functional polymers can be created, meeting the demands for novel materials in electronics and nanotechnology.
High Purity and Effectiveness
Methyl 6-bromonicotinate offers significant advantages in chemical synthesis. Its high purity level ensures consistent and reliable reaction results, minimizing the impact of unwanted by-products and impurities that could interfere with product isolation and purification. This feature is crucial for achieving optimal yields and efficient processes.
Versatility in Chemical Reactions
The multifunctionality of Methyl 6-bromonicotinate makes it a valuable reagent for a wide range of chemical transformations. Its reactive bromine atom readily participates in various substitution and coupling reactions, providing chemists with a powerful tool for constructing complex molecules. This broad applicability expands synthetic possibilities and aids in the development of new compounds.
Methyl 6-bromonicotinate is typically obtained from reputable chemical suppliers. These companies offer the compound both online and in-store. The price of this chemical may vary based on purity, quantity, and market demand. For bulk purchases, exploring volume buying options may significantly reduce costs. When selecting a supplier, prioritize those with a good reputation and transparent pricing to ensure product quality and customer satisfaction.
 |