Is n-butyllithium solid or liquid? n Butyllithium exists in clusters both in solid-state and solution. Clustering tendencies are common among organolithium compounds, where clusters are held together by nonlocalized covalent bonds between lithium and terminal carbon of the butyl chain. For n Butyllithium, clusters are tetrameric (in ether) or hexameric (in cyclohexane). The cluster forms a distorted cubane-type structure, with Li and CH2R groups at alternating vertices. A tetramer is equivalently described as intersecting tetrahedra Li4 and [CH2R]4. Bonding within the cluster resembles ethylborane but is more complex due to involvement of eight atoms. n Butyllithium exhibits electron-rich properties and high reactivity toward Lewis acids. The tetramer and hexamer of n Butyllithium are depicted in Figures 1 and 2, respectively:
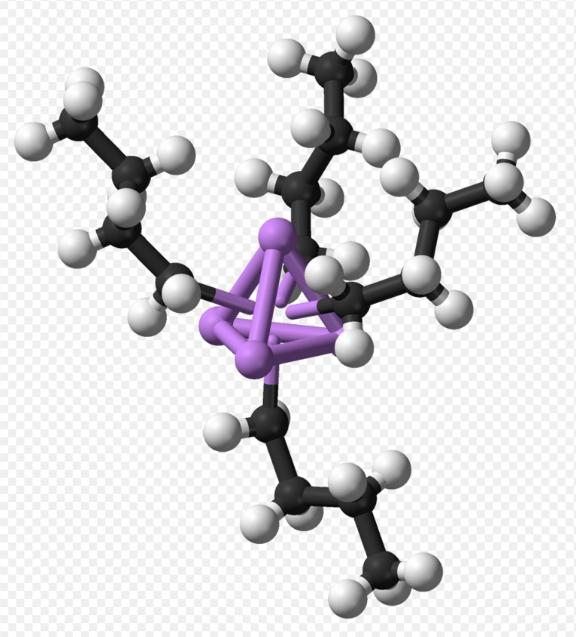
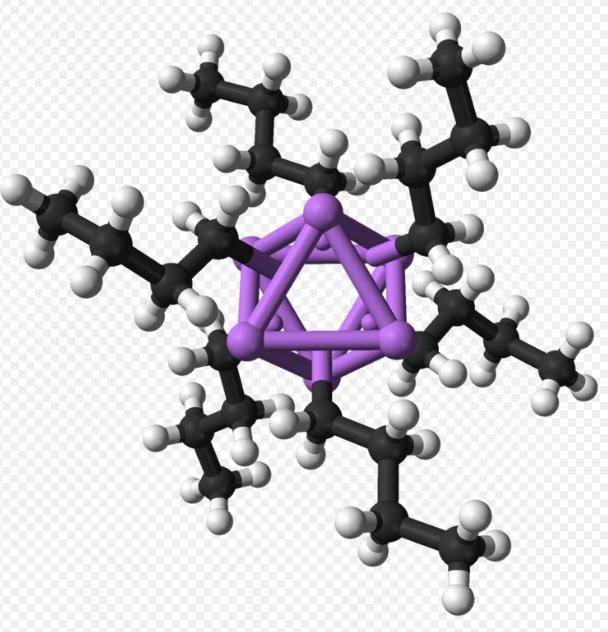
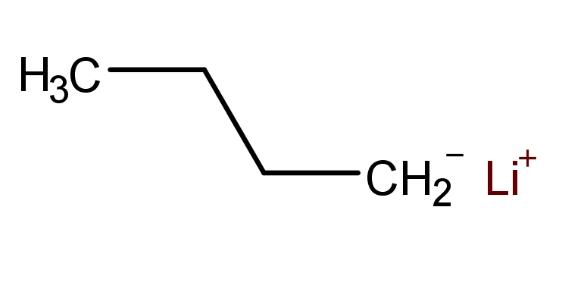
N Butyllithium, also known as n-BuLi, , is a significant organolithium compound. N Butyllithium cas no is 109-72-8. It typically exists as a colorless liquid but can also be powdery. n Butyllithium possesses the following physical properties:
N Butyllithium Molecular weight: 64.06 g/mol
N Butyllithium Melting point: -84°C
N Butyllithium Boiling point: 80-90°C (0.013 Pa)
N Butyllithium Density: 0.68 g/mL (20°C)
N Butyllithium Specific gravity: 0.765 (25°C)
N Butyllithium Solubility: Soluble in various organic solvents like pentane, hexane, benzene, and exothermic in water.
N Butyllithium Reactivity: Highly flammable in air, rapidly decomposing in water to lithium hydroxide and butane. Slow reaction with ether produces ethoxy lithium, butane, and ethylene. Slow decomposition at around 100°C, rapid at 150°C and above, mainly producing butene, butane, and lithium hydride.
N Butyllithium Caution: n Butyllithium is highly basic and flammable. Handling requires caution to avoid skin and eye contact and keep away from ignition sources.
n Butyllithium is a promising new product as an excellent anionic polymerization initiator, widely used in the synthesis of S-SBR, LCBR, MVBR, HVBR, SBS, SIS, KR-BDS, HBI, and liquid polybutadiene rubber. It is also a hydrocarbonation catalyst for synthesizing new chemicals used in rubber production, new medical antibacterial drugs, AIDS drugs, flavor synthesis, liquid crystal materials, and other fields.
n Butyllithium is a highly reactive pyrophoric compound, meaning it self-ignites upon contact with air. In laboratory and industrial settings, extreme caution must be exercised using inert gas techniques when handling n Butyllithium. This typically involves transferring reagents under a fume hood using syringes or cannulas, while purging original containers with inert gases like nitrogen or argon to prevent exposure to air. Personnel should wear appropriate personal protective equipment including gloves, goggles, and lab coats to prevent skin and eye exposure.
To maintain stability and prevent degradation, n Butyllithium must be stored under specific conditions. It should be kept in a cool, well-ventilated area away from heat sources and ignition hazards. Containers must be sealed and stored under an inert gas (typically nitrogen). Many suppliers offer pre-sealed n Butyllithium solutions under an inert gas. Minimize headspace when accessing n Butyllithium to reduce exposure to air. Consult safety data sheets (SDS) for specific storage recommendations provided by the manufacturer.
n Butyllithium is a highly valuable reagent in organic synthesis, but careful handling is essential to ensure safety. Understanding its applications, safety precautions, and regulatory considerations are crucial to preventing accidents and ensuring successful experiments. Responsible operation and adherence to safety guidelines are paramount when using n Butyllithium or other organolithium compounds. As organic synthesis technology advances, the application of n Butyllithium and other organolithium reagents is expected to expand, bringing more innovations and possibilities to the field of synthetic chemistry. By studying diligently and strictly following safety measures, we can better harness these organolithium compounds to drive scientific research forward.
[1] Liu, Z. J. Synthesis and Application of n-Butyllithium. Qilu Petrochemical, 2004, (S1): 56-58+61-10.
[2] https://en.wikipedia.org/wiki/N-Butyllithium
[3] https://pubchem.ncbi.nlm.nih.gov/
[4] https://enhs.uark.edu/_resources/documents/
[5] https://ehs.stanford.edu/reference/information-on-pyrophoric-compounds
 |
 |
 |