N-Isopropylaniline, with the CAS number 768-52-5 and molecular formula C9H13N, is a transparent pale yellow liquid with a sweet odor. It is utilized in dyeing acrylic fibers and in the production of other chemicals. N-Isopropylaniline is a metabolic product of propachlor under microbial action. Propachlor is an acylanilide herbicide used extensively to protect crops from weeds. Research indicates that propachlor may induce glutathione-dependent cytotoxic reactions and sister chromatid exchanges in human cells.

N-Isopropylaniline is an important intermediate in the production of pesticides, pharmaceuticals, dyes, and other products.
N-Isopropylaniline is a key intermediate in organic chemistry and is also used to synthesize other aromatic amines, such as aniline and phenylenediamine.
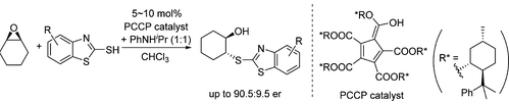
Chao Yuan et al. utilized a chiral pentacarboxycyclopentadiene-based Brønsted acid with N-Isopropylaniline as an amine additive to achieve enantioselective desymmetrization of 2-mercaptobenzothiazoles, obtaining ring-opening products with up to 90.5:9.5 er.
Eiji Ihara et al. studied the anionic polymerization of methyl methacrylate using lithium amides of various secondary amines, including N-Isopropylaniline, as initiators in toluene. The stereoregularity of the resulting polymethyl methacrylate (PMMA) depended on the type of secondary amine used, with N-Isopropylaniline and N-n-butylaniline yielding highly isotactic PMMA (91-93% mm).
Xiaohao Li et al. conducted intermolecular hydroaminomethylation and intramolecular cyclocarbonylation effectively on cobalt nanoparticle-supported gold catalysts. They used terminal olefins and N-Isopropylaniline in a common reaction pathway consisting of hydroformylation, imine formation, and hydrogenation to provide hydroaminomethylation products as a regioisomeric mixture.
N-Isopropylaniline has been used to study the metabolic mechanism of propachlor in Pseudomonas strains. Propachlor (2-chloro-N-isopropylacetanilide) was applied to organic soils in 1972 and 1975, and to onions (pre-emergence) in 1974 and 1975. Onions planted in May were harvested in September and analyzed in January. The analysis showed no residues of free propachlor or free N-Isopropylaniline in organic soils and onions, but N-Isopropylaniline was recovered after hydrolysis. Residues of this conjugated metabolite were found in organic soils 1 and 2 years after application. The amount of this metabolite increased in onion tissues with higher propachlor application rates and later stages of onion growth. N-Isopropylaniline has been identified as a metabolite of propachlor degradation, providing insights into the herbicide's breakdown pathways.
Research indicates that N-Isopropylaniline is a metabolite of isoproturon, with its adsorption behavior in soil varying with soil composition and land use. This information is crucial for understanding pesticide fate in the environment and developing strategies to reduce water pollution.
Jing Xuan et al. established a gas chromatography method for determining N-Isopropylaniline in workplace air. The method involves collecting air samples with a silica gel tube and desorbing them with acetone. The samples are then separated using a DB-WAX column and detected using a flame ionization detector. N-Isopropylaniline concentrations range from 1.40 to 665.0 μg/ml with good linearity. Sampling efficiency is 100%, with precision ranging from 96% to 99% and accuracy from 2.1% to 7.0%. The minimum detectable concentration is 0.056 mg/m³ (for a 7.5 L air sample). The method meets analytical requirements and is suitable for determining N-Isopropylaniline in workplace environments.
Avoid contact with skin and eyes. Prevent dust and aerosol formation. Follow special instructions before use. Provide appropriate exhaust ventilation where dust is formed. Wear suitable protective clothing and gloves.
Store separately from food and feed. Keep cool. Store in a well-ventilated area. The storage area should be cool, ventilated, dry, and fireproof. Keep separate from oxidizers and strong acids.
 |
 |