-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

Fluorine, heterocyclic, and chiral compounds are the three main features of modern drug and pesticide synthesis. Among these, pyridine-based fluorine compounds are the most commonly used due to current synthesis technology. Pyridine fluorinated compounds combine fluorine, nitrogen-containing heterocyclic, and chiral properties. In recent years, compounds with these characteristics have been widely studied and applied in the fields of medicine and pesticides. Pentafluoropyridine is an important intermediate in both industries and has significant research value. Fluorine, being highly electronegative, can reduce the oxidation rate of materials, making fluorine-substituted organic materials highly valuable in aerospace and biomedical applications.
Pentafluoropyridine formula: C5F5N; Molecular weight: 169; Boiling point: 84℃; Structure:
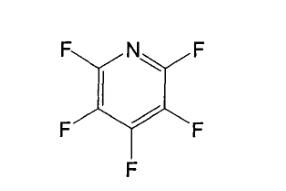
Pentafluoropyridine structure
Lido et al. optimized the ground state geometric structure of Pentafluoropyridine using B3LYP, M062X, and SA-CASSCF(8,8) methods with the 6-311G* basis set. The results indicated that the ground state geometric structures obtained under different methods were similar, all being planar structures with C2v symmetry. The C-C and C-F bond lengths in the S0 state of Pentafluoropyridine were 1.38-1.39Å and 1.31-1.33Å, respectively, while the C-N bond length was 1.31Å, slightly shorter than the C-C bond length.
Synthesis of Highly Functionalized Heterocycles and Macrocycles: Pentafluoropyridine is a multifunctional “building block” for synthesizing various substituted pyridines and macrocyclic derivatives with potential applications in life sciences and supramolecular chemistry.
Synthesis of Pyridine Derivatives: H. Benmansour and colleagues synthesized a series of pyridine derivatives using Pentafluoropyridine as a raw material, utilizing nucleophilic aromatic substitution and palladium-catalyzed coupling reactions.
Peptide System Modification: Pentafluoropyridine, as a perfluoroaromatic reagent, has proven to be a highly reactive electrophilic reagent, enabling SNAr arylation reactions with peptide side chains under mild conditions, offering enhanced proteolytic stability in drug-related peptides.
Amide Synthesis: W. D. G. Brittain reported the application of Pentafluoropyridine in carboxylic acid deoxyfluorination for synthesizing acyl fluorides, which can be further used in one-pot amide bond formation.
Applications in Polymers: Johncock, Hewins, and Cunliffe were the first to report the use of Pentafluoropyridine in polymer synthesis, aiming to develop materials with enhanced structure and oxidative stability for aerospace applications.
Gas Chromatography-Mass Spectrometry Analysis: Sanka N. Atapattu et al. developed a simple, low-cost method using Pentafluoropyridine as a derivatizing reagent to analyze endocrine-disrupting chemicals using solid-phase analytical derivatization technology.
Due to its unique chemical properties and extensive application potential, Pentafluoropyridine has become an indispensable compound in the pharmaceutical and material science fields. Its excellent performance in catalysis and synthesis reactions continues to drive technological advancements. For more information on Pentafluoropyridine and supplier details, visit Guidechem.
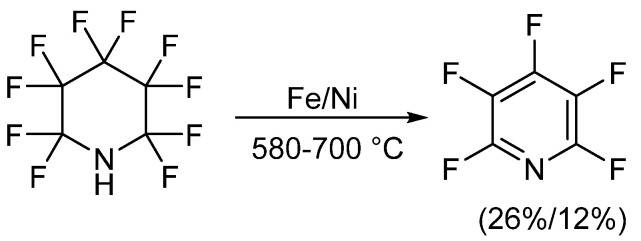 |
 |