-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here


Phthalazine, also known as 2,3-diazaphenalene, is an important nitrogen-containing heterocyclic compound. The phthalazine unit serves as a fundamental structural component in many biologically active organic compounds, gaining increasing attention in pharmaceuticals, agrochemicals, and functional materials. The phthalazine structure is shown below. DNA repair enzyme (PART) is a cellular inhibitor that can inhibit DNA repair in tumor cells. Desloratadine hydrochloride, with antihistamine and anti-inflammatory properties, can treat seasonal allergic rhinitis. Watarlanin has antitumor activity. Additionally, phthalazine compounds also exhibit excellent performance in luminescent materials and fluorescent probes.

Phthalazine is a heterocyclic organic compound with the molecular formula C8H6N2. It is a pyrazine related to phthalic anhydride, and it exists as an isomer with other naphthalene-based compounds, including quinoxaline, cinnoline, and quinazoline. The phthalazine structure can be viewed as a naphthalene molecule where carbons 2 and 3 are replaced by nitrogen atoms. Phthalazine consists of a fused benzene ring and a pyrazine ring, containing nitrogen heterocycles with hydrazine (two adjacent nitrogen atoms), playing an essential role in organic synthesis. Phthalazine-1(2H)-one and 2,3-dihydrophthalazine-1,4-dione are two important functional forms of the phthalazine structure. The structure of phthalazine is as follows:

Phthalazine structure
The spectral properties of nitrogen-containing aromatic compounds like pyrazine and phthalazine differ significantly from their isomers, such as pyridine and quinoline. The triplet state characteristics of the latter have been extensively studied using various spectroscopic techniques, including ODMR. Due to the non-phosphorescent nature of pyrazine under any conditions, the triplet state of phthalazine remains unknown. Pyrazine and phthalazine produce very good TREPR signals in rigid media at low temperatures.
The triplet state properties of phthalazine are highly dependent on the environment and temperature. Nitrogen hyperfine splitting was not observed in benzyl and 1,4-dichlorobenzene hosts, but it was clearly observed in biphenyl and anthracene. The sublevel order changes from Z>Y>X in benzyl to Z>X>Y in anthracene. Hyperfine splitting and ZFS analysis indicate that at 4.2K, the T state features approximately 60% ππ* and 40% nπ*. nπ*‐ππ* vibronic mixing is accompanied by out-of-plane distortions of the molecule. Yamauchi and others indeed detected this distortion through time-resolved ENDOR work.
In 2001, Napoletano and others derivatized phthalic anhydride, using methanol as a solvent under acidic conditions to condense with hydrazine, obtaining phthalazine ketone derivatives with almost quantitative yields. Subsequently, the carbonyl group was chlorinated and underwent a coupling reaction under palladium catalysis, forming 1,4-substituted phthalazine derivatives (as shown in the figure).

In 2010, Hegab and others used phthalic anhydride in a Friedel-Crafts reaction with aromatic hydrocarbons to generate ortho-aroylbenzoic acids, which were then condensed with hydrazine to obtain 4-substituted phthalazine ketone compounds. Through multiple reactions, a series of highly active pharmaceutical molecules were synthesized, with anti-inflammatory effects surpassing indomethacin (as shown in the figure).
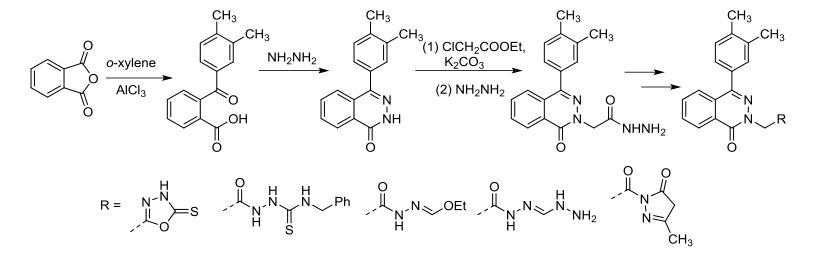
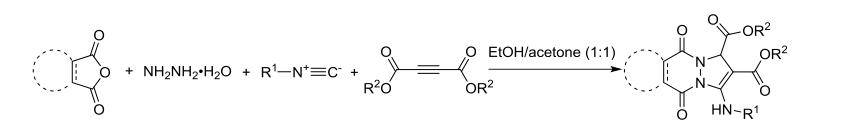
In 2012, Shaabani and his team discovered that phthalic anhydride, hydrazine, isocyanides, and butyne-2,3-dicarboxylates could undergo a four-component reaction in a mixed solvent of ethanol and acetone at room temperature, yielding highly substituted phthalazine derivatives with moderate yields. The researchers suggested that the formation of 1,4-dipole from isocyanides and butyne-2,3-dicarboxylates drives this reaction. This dipole undergoes nucleophilic addition with phthalazine ketone formed in situ from phthalic anhydride and hydrazine, completing the reaction to yield the final product (as shown in the figure).
The synthesis of phthalazine derivatives can also start from existing phthalazine cyclic compounds, further modifying and extending their structure using the nucleophilic properties of nitrogen atoms. In 2008, Bazgir and others reported a three-component reaction where phthalazine dione, malononitrile or cyanoacetic ester, and aromatic aldehydes were heated in ionic liquids, successfully synthesizing compounds with a pyrazole[1,2-b]phthalazine dione structure. The authors noted that aromatic aldehydes first condensed with malononitrile or cyanoacetic ester to produce α, β-unsaturated carboxylic acid derivatives. Phthalazine dione then underwent 1,4-addition, and the other nitrogen atom in phthalazine ketone reacted nucleophilically with cyano groups to yield the product (as shown in the figure).

When phthalazine has halogens at the 1- or 4-position, it exhibits properties similar to acyl chlorides, allowing for nucleophilic substitution reactions. Thus, using nucleophiles to react with 1- or 4-chlorophthalazine is a common method for preparing phthalazine derivatives. In 2004, Lebsack and other researchers reacted with 1,4-dichlorophthalazine to form fused-ring structures and synthesized potential anticonvulsant drug molecules through multiple steps, all containing the phthalazine structure (as shown in the figure).

[1] Chen Chen. Research on green synthesis methods of phthalazines, bisindoles and 3-substituted indoles containing nitrogen heterocyclic compounds[D]. Liaoning: Liaoning Normal University, 2020.
[2] Luo Mingjian. Study on the addition reaction of phthalazine salts with terminal alkynes[D]. Heilongjiang: Harbin Institute of Technology, 2019.
[3] Zheng Xuanming. Synthesis of phthalazine-based α-aminophosphonates[D]. Heilongjiang: Harbin Institute of Technology, 2018. DOI:10.7666/d.D01587313.
[4] Zhao Meimei, Tong Hongjuan, Du Mo. Research progress on the synthesis and activity of phthalazine derivatives [J]. Fine and Specialty Chemicals, 2022, 30(4): 15-19. DOI: 10.19482/j.cn11-3237.2022. 04.03.
[5]https://en.wikipedia.org/wiki/Phthalazine
[6]Sangshetti J, Pathan S K, Patil R, et al. Synthesis and biological activity of structurally diverse phthalazine derivatives: A systematic review[J]. Bioorganic & Medicinal Chemistry, 2019, 27(18): 3979-3997.
 |
 |
 |