-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

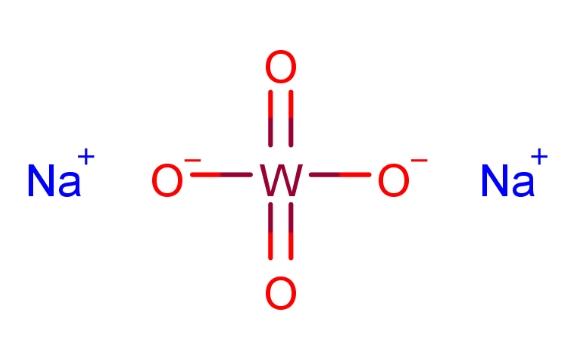
Sodium tungstate is an inorganic compound with the chemical formula Na2WO4. It appears as colorless or white orthorhombic crystals, usually glossy flaky crystals or powder. Sodium tungstate is soluble in water, forming a slightly alkaline solution, but it is insoluble in ethanol and only slightly soluble in ammonia. Sodium tungstate weathers in the air and loses crystal water when heated to 100℃, converting into anhydrous sodium tungstate. In strong acids (except hydrofluoric acid), sodium tungstate generates insoluble yellow tungstic acid. It also reacts with phosphoric acid or phosphates to form phosphotungstic heteropoly acid complexes and with organic acids like tartaric acid, citric acid, and oxalic acid to form corresponding organic acid complexes.
The solubility of sodium tungstate is as follows:
(1) Sodium tungstate solubility in water:96.9 g/100 mL (100 ℃)
Insoluble in alcohol, acid
The solubility of sodium tungstate (Na2WO4) in molten sodium hydroxide (NaOH) baths and its dependence on water vapor pressure was measured as part of ongoing research using molten NaOH for a new tungsten (W) recovery process. First, the nature of the compounds in the equilibrium solid phase with saturated Na2WO4 in the molten NaOH bath was confirmed by adding an excess of tungsten oxide as a tungstate ion source. X-ray diffraction analysis indicated that anhydrous Na2WO4 is the solid phase in equilibrium with the liquid phase of molten NaOH saturated with sodium tungstate. The solubility of anhydrous sodium tungstate was then measured under various water vapor pressures. Results showed that the solubility of anhydrous sodium tungstate increases sharply with rising water vapor pressure and bath temperature, with the mole fraction increasing from 0.06 to 0.17. This value is much higher than previously reported values for molten nitrates and nitrites.
This experiment studied the solubility of sodium tungstate in molten NaOH, with the experimental results summarized as follows:
Confirmed that the equilibrium solid phase compound at 723 K in the Na2WO4–NaOH–H2O system is anhydrous sodium tungstate.
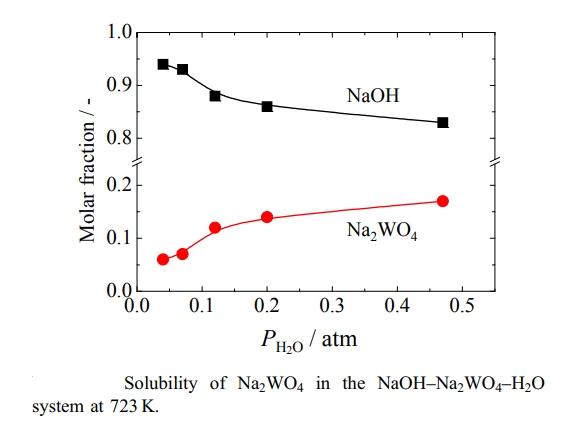
The solubility of sodium tungstate increases with the increase in water vapor pressure and bath temperature.
The obtained solubility range was 1.5 to 5.2 mol-Na2WO4/kg-melt, far exceeding the solubility of nitrate and nitrite melts.
Pengge Ning et al. determined the solubility of Na2MoO4·2H2O and Na2WO4·2H2O in the Na2MoO4–Na2WO4–Na2SO4–H2O system. The solubility of dihydrate sodium tungstate in (0.1–1.0) mol·kg?1 sodium sulfate solution (temperature range (293.15 to 343.15) K) was measured using the dynamic method. The results indicated that the solubility of dihydrate sodium tungstate increases with increasing temperature and decreases with increasing sodium sulfate concentration. The dissolution of dihydrate sodium tungstate is an endothermic process, and considering the role of sodium sulfate, the common ion effect of Na+ is more significant.
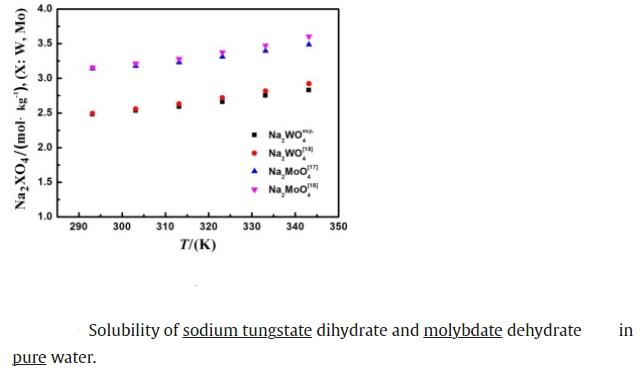
The solubility of sodium tungstate is significantly influenced by temperature. Generally, the solubility of sodium tungstate in water increases with the rising solvent temperature. This trend is due to the endothermic nature of the dissolution process for many salts, including sodium tungstate. As the temperature rises, the additional thermal energy helps to overcome the lattice energy of the solid, promoting the dissolution of the salt into its constituent ions. For sodium tungstate, this means that more of the salt can dissolve in water under warmer conditions, leading to higher concentrations in the solution.
The pH value of the solution can also affect the solubility of sodium tungstate. While sodium tungstate is generally considered soluble in water over a wide pH range, extreme pH conditions may impact its solubility. For example, very acidic or alkaline environments may lead to the formation of insoluble tungstate compounds. Maintaining the pH within an appropriate range is essential to ensure complete dissolution of sodium tungstate and to prevent unwanted precipitation.
The choice of solvent and the specific conditions under which solubility is tested also play a crucial role. Sodium tungstate is most commonly dissolved in water, with its solubility affected by temperature and pH as mentioned earlier. However, other solvents (like alcohol or organic solvents) may not effectively dissolve sodium tungstate due to differences in polarity and solvent-solute interactions. Additionally, factors like ionic strength of the solution, presence of competing ions, and even the stirring rate can influence the dissolution process. Understanding these variables is essential for optimizing the solubility of sodium tungstate in various industrial and laboratory applications.
Sodium tungstate is most easily dissolved in water. The most common practice is to use deionized or purified water as the solvent. Sodium tungstate has high solubility in water, especially in warm water. If higher solubility is required, warm water can be considered. Stirring can accelerate the dissolution process when dissolving sodium tungstate. Continuous stirring with a stirring rod or magnetic stirrer helps sodium tungstate dissolve evenly. For large quantities of sodium tungstate, the stirring time may need to be extended.
If there are other high concentrations of ions in the solution, they may affect the solubility of sodium tungstate. It is important to maintain the concentration of other ions in the solution within an appropriate range to avoid negatively impacting the dissolution process.
Tungsten and molybdenum are rare refractory metals. They are both high-temperature resistant and excellent conductors, making them important national strategic resources. The recovery and purification of these two metals often occupy a central position in hydrometallurgy processes, attracting considerable attention from researchers. Due to their respective advantages, many methods have been employed to collect tungsten and molybdenum, such as precipitation, solvent extraction, ion exchange, and adsorption. Among these methods, precipitation and solvent extraction are more popular and widely applied in certain industries. However, any treatment method inevitably has additional drawbacks or deficiencies.
The greatest challenge in recovering these two metals comes from their similar properties. Tungsten and molybdenum belong to the same subgroup of elements and have very similar chemical and physical properties. Additionally, tungsten and molybdenum often coexist in the same ore or secondary resource, increasing the difficulty of extraction. Furthermore, the aqueous chemistry of tungsten and molybdenum is complex. In tungsten- or molybdenum-containing aqueous solutions, acid polymerization always results in the formation of isopoly acids. In tungsten-molybdenum-containing aqueous solutions, hydrolysis and polymerization reactions not only produce isopoly acids but also heteropoly acids. Among all factors, pH and metal concentration are the main determinants of metal species.
Solvent extraction is considered a promising method due to its high product purity, simple operation, and high separation efficiency. Various methods of recovering tungsten or molybdenum using solvent extraction based on different mechanisms have been studied. Therefore, it is important to study the distribution and thermodynamic properties of tungsten and molybdenum species in aqueous solutions under different conditions.
[1]Oishi T, Yaguchi M. Solubility of sodium tungstate in molten sodium hydroxide[J]. Electrochemistry, 2018, 86(2): 61-65.
[2]https://www.sciencedirect.com/science/article/abs/pii/S002196141600032X
[3]https://baike.baidu.com/item/%E9%92%A8%E9%85%B8%E9%92%A0
[4]https://en.wikipedia.org/wiki/Sodium_tungstate
[5]https://www.sciencedirect.com/topics/chemistry/sodium-tungstate
[6]Central South University. A method for preparing tungsten products from crude sodium tungstate solution: CN201810362727.2[P]. 2020-07-31.
 |
 |
 |