TSTU chemical name N, N, N, N-Tetramethyl-O-(N-succinimidyl)uronium tetrafluoroborate, is a widely used reagent for amide bond formation in organic synthesis. Tstu chemical formula is C9H16BF4N3O3, and it is identified by CAS number 105832-38-0.
TSTU has a molecular formula of C9H16BF4N3O3 and a molecular weight of 301.05 g/mol. The structure of this compound is complex, featuring several functional groups. TSTU consists of two main components: the cation and the anion. The cation part is (2-(2,5-dioxopyrrolidin-1-yl)-1,1,3,3-tetramethylurea), with a central nitrogen atom bonded to four methyl groups (CH3) and linked to a five-membered ring (succinimide ring) containing two adjacent carbonyl groups (C=O). The anion is the tetrafluoroborate ion (BF4-), with a tetrahedral structure composed of a central boron atom and four fluorine atoms. The positive charge of the cation is balanced by the negative charge of the anion, forming a stable ionic compound.
TSTU is a versatile coupling reagent commonly used in organic chemistry, particularly in peptide synthesis. The formation of amide bonds is a crucial step in synthesizing various biologically significant organic compounds, notably peptides and peptide-like compounds, though they are also found in low-molecular-weight amides, β-lactams, polyamides, benzodiazepines, diketopiperazines, and hydrazides. Peptide coupling agents are typically used to activate carboxylic acids for amide bond formation.
In recent years, numerous such coupling agents have appeared in the literature, improving upon older methods. Among today's preferred peptide coupling agents are ammonium/urea derivatives, valued for their higher efficiency and lower tendency to racemize amino acids or peptide residues. These salts are usually prepared by reacting XOH-type compounds with urea (e.g., tetramethylurea (TMU)) to form halide salts. Common ammonium/urea salts include 1-hydroxybenzotriazole derivatives (TBTU and HBTU), 7-azabenzotriazole derivatives (TATU and HATU), 4-hydroxy-3,4-dihydro-1,2,3-benzotriazine-4-one derivatives (TDBTU and HDTU), 1-hydroxy-1,2-dihydro-2-pyridone derivatives (TPTU and HPTU), and 1-hydroxy-succinimide derivatives (TSTU and HSTU), among others. These compounds are widely available on the market.
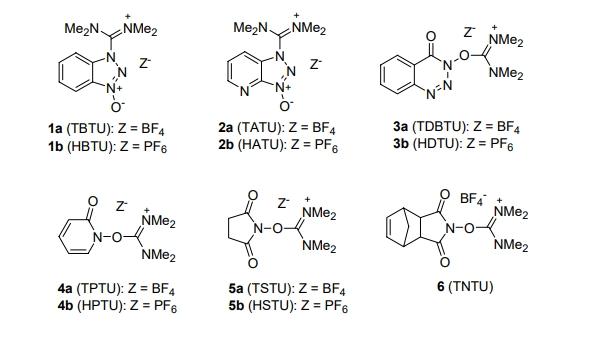
TSTU is highly favored in organic synthesis due to several advantages:
TSTU (N, N, N, N-Tetramethyl-O-(N-succinimidyl)uronium Tetrafluoroborate) is highly regarded in chemical reactions for its excellent reaction characteristics. As an efficient coupling reagent, TSTU provides superior reaction selectivity and high yield in various chemical syntheses. Its stability and reactivity make it an indispensable tool in synthetic chemistry, especially in scenarios requiring precise control of reaction conditions. For further information about TSTU or to purchase this high-performance reagent, visit the Guidechem platform. On Guidechem, you can find detailed information about TSTU and make a purchase. Guidechem offers reliable supplier information, ensuring the quality and reliability of the product supply.
[1]https://www.medchemexpress.cn/tstu.html
[2]Chinchilla R, Dodsworth D J, Nájera C, et al. Uronium salts from polymeric N-hydroxysuccinimide (P-HOSu) as new solid-supported peptide coupling reagents[J]. Arkivoc, 2003, 10: 41-47.
[3]https://pubchem.ncbi.nlm.nih.gov/compound/O-(N-Succinimidyl)-1,1,3,3-tetramethyluronium tetrafluoroborate
[4]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8948954/
 |
 |