-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

Recently, Boundless, a clinical-stage oncology company focused on advancing transformative therapies for patients with difficult-to-treat cancer caused by oncogene amplification, announced updates to its ecDNA-targeted therapy (ecDTx) pipeline and its executive leadership team.
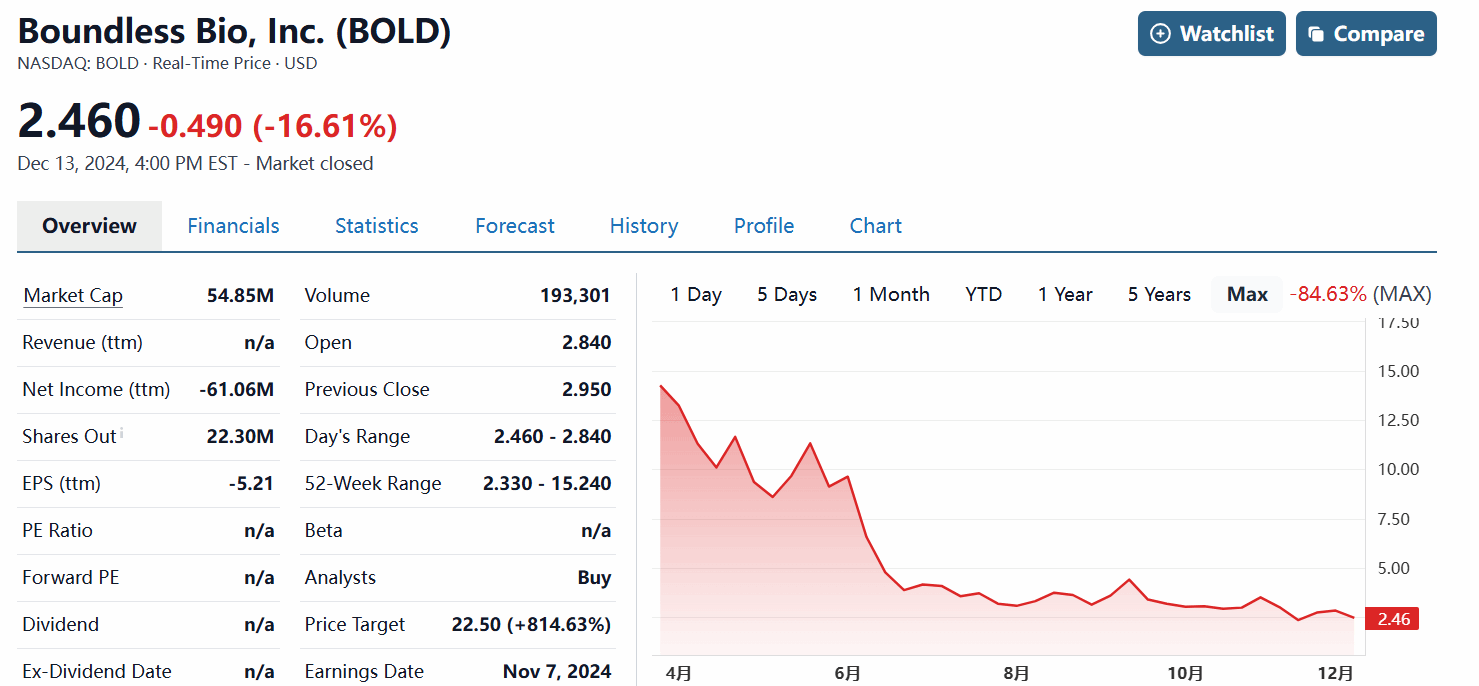
It is reported that Boundless went public with $100 million in funding a year ago, but its market capitalization has since halved:
Boundless announced the following updates to its novel ecDTx pipeline targeting a range of aggressive and difficult-to-treat cancer indications:

Boundless official website pipeline
Boundless also announced that Chief Medical Officer (CMO) Dr. Klaus Wagner and Chief Business Officer (CBO) Neil Abdollahian will be leaving the company at the end of December and early January, respectively. Dr. James L. Freddo, currently a consultant to Boundless with nearly 30 years of clinical leadership experience in the biopharma industry, will serve as interim CMO while the company searches for a permanent hire. Dr. Freddo's extensive background includes serving as CMO and board member at Ignyta, Inc., CMO at Anadys Pharmaceuticals, and Vice President of Oncology Clinical Development at Pfizer. Boundless does not intend to appoint a new CBO at this time.
“After a comprehensive evaluation of the new clinical data for BBI-825 and anticipated development costs, particularly in the highly competitive BRAF V600E and KRAS G12C mutation cancer treatment landscape, we have made the decision not to advance the STARMAP trial. We thank the team members, patients, and investigators who contributed to the BBI-825 program,” said Zachary Hornby, President and CEO of Boundless Bio. “With this strategic decision, we will prioritize resources on BBI-355, which remains on track to deliver preliminary clinical proof-of-concept data in 2025, as well as our novel ecDTx 3 program, in which we have made substantial preclinical progress and expect to nominate a development candidate by mid-2025. It also extends our operational runway into 2027, well beyond the anticipated milestones for BBI-355 and ecDTx 3.”
Mr. Hornby added, “At Boundless, we are focused on improving outcomes for patients with oncogene-amplified cancers by tackling complex challenges in cancer biology. I want to sincerely thank Klaus and Neil for their significant contributions over the past few years and wish them all the best in their next professional chapters. Looking ahead, we remain committed to delivering meaningful results for patients and other stakeholders, and we look forward to sharing our progress in 2025.”
https://www.globenewswire.com/news-release/2024/12/12/2996511/0/en/Boundless-Bio-Announces-Pipeline-and-Leadership-Updates.html
https://boundlessbio.com/what-we-do