Adipic acid is an important organic compound with two carboxyl groups. In practical applications, we often need to consider the solubility of adipic acid in different solvents to better utilize this compound in fields such as chemical production and pharmaceutical preparation. The solubility of adipic acid is influenced by various factors, including solvent polarity, temperature, and pressure. By exploring the solubility of adipic acid in various solvents, we can gain insight into its dissolution mechanism, providing valuable references for selecting solvents and optimizing production processes.
Adipic acid, a valuable raw material, is used in the production of nylon-66, fiber lubricants, plasticizers, and food additives. It is typically manufactured through the oxidation of cyclohexanone/cyclohexanol, hydrogenation of phenol, or more commonly, oxidation of cyclohexane. The crude product of oxidation reactions contains a range of impurities that must be purified. Recrystallization is a purification method commonly employed. Apart from its solubility in water and ethanol, adipic acid is virtually insoluble. Additionally, a small amount of adipic acid needs to be separated from acidic washing water, a byproduct of air oxidation of cyclohexane. Therefore, understanding the solubility of adipic acid is crucial.
Is adipic acid insoluble? Adipic acid exhibits moderate solubility in water. While some studies may claim it to be insoluble, it is actually soluble to some extent. The solubility is as follows:
14 g/L at 10°C
24 g/L at 25°C
1600 g/L at 100°C
Adipic acid forms hydrogen bonds with water molecules due to its carboxyl groups (COOH). However, the presence of the carbon-hydrogen chain (CH2) disrupts extensive hydrogen bonding, limiting its solubility compared to highly soluble ionic compounds.
As shown, solubility significantly increases with temperature. Higher temperatures increase the molecular kinetic energy, making adipic acid more capable of overcoming the attractive forces holding it together and dissolving in water.
Adipic acid is a weak acid. In highly acidic solutions (low pH), the equilibrium favors the undissociated form, reducing solubility in water. Conversely, slightly alkaline solutions (high pH) may favor the dissociated ion form, potentially increasing solubility.
Adipic acid is a crucial component in nylon synthesis. Its moderate solubility allows for controlled polymerization reactions in aqueous environments.
Due to its limited solubility, adipic acid is used as an acidity regulator in some foods. Its slow release aids in maintaining stable pH levels.
The solubility curve of adipic acid can be tailored for specific drug delivery applications. For example, controlled-release formulations may leverage its temperature-dependent solubility.
Adipic acid is soluble in dimethyl sulfoxide (DMSO). While the exact solubility may vary with temperature and other factors, adipic acid exhibits good solubility in DMSO.
FAN Lihua et al. measured the solubility data of adipic acid dissolved in six pure solvents, namely cyclohexanone, cyclohexanol, acetic acid, N,N-dimethylformamide, N,N-dimethylacetamide, and dimethyl sulfoxide (DMSO), using a laser detection system, with temperatures ranging from 293.15K to 353.15K. All data were regressed using λh, NRTL, Wilson, and modified Wilson models. The results, with x representing the mole fraction of adipic acid, are as shown in the table below:
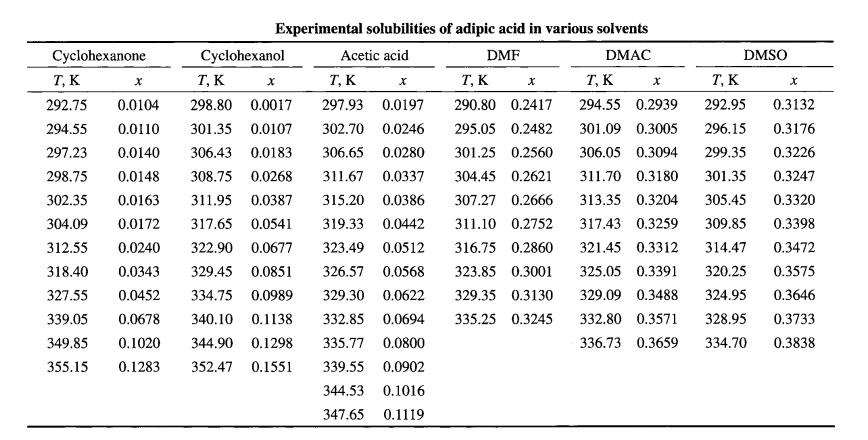
This study compared the solubility of adipic acid in six solvents including cyclohexanone, cyclohexanol, and acetic acid. Among the different solvents, DMSO exhibited the highest solubilizing capacity while cyclohexanone showed the lowest. The solubility of adipic acid in DMSO, DMAC, and DMF was approximately 20 times higher than that in cyclohexanone, cyclohexanol, and acetic acid. With increasing temperature, the solubility of adipic acid rises slowly, suggesting that these six solvents may not be suitable for cooling adipic acid recrystallization.
The differences in solubility of adipic acid in the six solvents are primarily attributed to their structures. The lower solubility of adipic acid in cyclohexanone and cyclohexanol may be due to structural incompatibility, as adipic acid has a chain structure while cyclohexanone and cyclohexanol have cyclic structures. The self-association of acetic acid reduces its solvating ability, hence the lower solubility of adipic acid in acetic acid compared to the other solvents.
Adipic acid can dissolve in sodium bicarbonate, sodium hydroxide, and concentrated sulfuric acid but not in hydrochloric acid. The solubility of the substance in pure water depends solely on temperature. However, adding some strong acids (such as hydrochloric acid) will decrease the solubility of weak acids (like adipic acid).
It reacts with sodium bicarbonate (baking soda) in water. This reaction is due to adipic acid being a diacid and sodium bicarbonate being a base. The reaction yields sodium adipate, water, and carbon dioxide gas. Sodium adipate is more soluble in water than the original adipic acid. Hence, although adipic acid cannot directly dissolve in sodium bicarbonate, its solubility in the solution can be increased through a chemical reaction. This reaction finds application in various processes:
Water-soluble effervescent disinfectant tablets reported by Ulick Stafford et al. contain dichloroisocyanuric acid esters, such as anhydrous sodium dichloroisocyanurate. The tablets also include alkaline metal hydrogen carbonates such as sodium bicarbonate, aliphatic carboxylic acids such as adipic acid, and alkali desiccants such as sodium carbonate. The tablets contain masking agents, particularly ethyl acetate or L-methyl ketone, in an amount sufficient to mask the taste and/or odor characteristics of dichloroisocyanuric acid esters. The addition of aliphatic carboxylic acids to the formulation, when reacted with alkaline metal hydrogen carbonates and other carbonates, releases carbon dioxide bubbles, thus promoting the effervescence breakdown of the tablets. Adipic acid is preferred as the aliphatic carboxylic acid. Its advantages include non-hygroscopicity, aiding in maintaining the integrity and stability of the finished formula, and when added to water, the effervescence reaction slows down most of the chlorine dissolution in the solution. It also has lubricating properties, aiding in the tabletting process.
Sodium bicarbonate is a weak base. When it reacts with adipic acid (a weak acid), it helps to neutralize acidity. This is useful in various scenarios.
Succinic acid (C4) has higher water solubility than adipic acid (C6) because it is smaller in size, offering more available sites for hydrogen bonding with water.
The water solubility of decanedioic acid (C10) and higher-chain dicarboxylic acids gradually decreases compared to adipic acid. This is because the increased hydrophobicity (water repellency) of longer carbon chains outweighs the effect of the two carboxylic acid groups.
Adipic acid generally has lower solubility in water compared to short-chain alcohols like ethanol or methanol. This is because alcohols can form more hydrogen bonds with water due to the presence of oxygen and hydroxyl groups (OH) in the carboxylic acid group (COOH).
Esters formed from adipic acid, such as dimethyl adipate, usually have lower solubility in water than adipic acid itself. This is because the ester bonds, which are less polar, replace the carboxylic acid groups, reducing the hydrogen bonding capability with water.
Increasing the length of the carbon chain (such as from succinic acid to decanedioic acid) increases the molecule's hydrophobicity, leading to decreased water solubility.
The presence of carboxylic acid groups (COOH) allows for hydrogen bonding with water. However, the number of available bonding sites and the competition from hydrophobic chains affect overall solubility. Shorter chains and chains with more hydrogen bonding sites (like succinic acid) are more easily soluble in water.
Additional functional groups are present. Replacing carboxylic acid groups with ester bonds, as in dimethyl adipate, reduces hydrogen bonding potential, decreasing water solubility.
Overall, the interplay between chain length, hydrogen bonds, and other functional groups determines the solubility of adipic acid relative to other dicarboxylic acids and similar compounds.
By exploring and researching the solubility of adipic acid in various solvents, we gain insight into the solubility properties of this organic compound in different environments. The solubility of adipic acid is influenced by various factors, including solvent properties, temperature, pressure, and others. In practical applications, we need to choose suitable solvents based on specific conditions to achieve effective dissolution and utilization of adipic acid.
[1] https://chemistry.stackexchange.com/questions/171550/does-decreasing-ph-decrease-solubility-of-adipic-acid
[2] https://www.chegg.com/homework-help/questions-and-answers/circle-solvents-expect-compound-would-soluble--example-adipic-acid-soluble-aqueous-sodium--q25328776
[3] https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/adipic-acid
[4] https://patents.google.com/patent/WO2010113144A2/en
[5] https://www.sciencedirect.com/science/article/abs/pii/S1004954107600421
 |
 |
 |