Adipic acid is a crucial organic compound with diverse applications in industries such as chemicals, pharmaceuticals, and food. It is synthesized through various methods, each with its unique advantages and applicability. This article aims to explore the preparation methods of adipic acid, delving into its synthesis principles and reaction mechanisms. By providing a detailed overview of the manufacturing process of adipic acid, we gain better insights into its production technology, offering vital references for research and applications in relevant fields.
Adipic acid (butanedioic acid, C6H10O4) is a white crystalline powder belonging to the class of C6 - straight-chain dicarboxylic acids. It is one of the most widely used chemicals globally. Adipic acid is sparingly soluble in water but soluble in alcohols and acetone.
Adipic acid was first obtained by Dieterle and his colleagues in 1884 through the oxidation of castor oil.
Adipic acid, whose name originates from the Latin word "adeps" meaning fat, is a product of fat oxidation (lipid peroxidation). Almost 90% of adipic acid consumption is related to nylon (nylon-6.6) production through its condensation with 1,6-hexamethylenediamine. Adipic acid is used in the manufacture of plasticizers, lubricants, and polyester polyols for polyurethane systems. The acid and its derivatives are also employed in the food industry (as a gelling agent, acidulant, flavoring agent, leavening agent, and buffering agent), as well as in the preparation of pesticides, dyes, textile auxiliaries, germicides, and drugs (such as cephalosporin intermediates).
Bouveault and R. Locquin reported that adipic acid can be produced by oxidizing cyclohexanol. Presently, the most common manufacturing process involves the oxidation of cyclohexanol-cyclohexanone mixture with nitric acid (HNO3), known as KA (ketone-alcohol) oil.
Adipic acid has two primary sources:
This is the most common method of obtaining adipic acid. It is produced by the oxidation of a mixture of cyclohexanone and cyclohexanol (referred to as KA oil) with nitric acid. This process involves multiple steps but essentially converts cyclohexane ring into a straight chain with two carboxylic acid groups.
Where is adipic acid found naturally? Adipic acid can be found in small amounts in some foods such as beets and sugarcane. However, this is not a practical method for obtaining large quantities of adipic acid.
Adipic acid is produced by the oxidation of a mixture of cyclohexanone and cyclohexanol, known as KA oil, with nitric acid serving as the oxidant. This process involves multiple steps. In the early stages of the reaction, cyclohexanol is converted into ketone, releasing nitrous acid. Cyclohexanone is then nitrosated. The byproducts of this method include glutaric acid and succinic acid. Nitric acid reacts with adipic acid in a molar ratio of approximately one to one, also producing nitric oxide through intermediates of nitric acid.
The early commercial processes for adipic acid involved the two-step air oxidation of cyclohexane. Cyclohexane's low conversion to cyclohexanol-cyclohexanone mixture was then subjected to high conversion air oxidation to adipic acid. However, all large-scale production today involves the oxidation of cyclohexanol, cyclohexanone, or a mixture of both (KA oil) with nitric acid. The differences between commercial processes primarily lie in the manufacturing of KA oil. The six carbon atoms of the adipic acid skeleton usually come from benzene, hydrogenated to cyclohexane, or phenol, hydrogenated to cyclohexanol. Cyclohexane is then oxidized to KA oil in the air. Over the past 20 years, there has been a shift towards the lower-cost cyclohexane process. Significant research has been conducted since the early 1980s on synthesizing adipic acid from butadiene and carbon monoxide.
The second step of the conventional nitric acid oxidation of cyclohexanol is developed by DuPont in the late 1940s, involving the oxidation of cyclohexanol, cyclohexanone, or a mixture of both with nitric acid. The yield of adipic acid is over 90%. The major byproducts are carbon dioxide, nitrogen oxides, and some low molecular weight dicarboxylic acids. Some byproducts are formed from impurities in the starting KA oil.
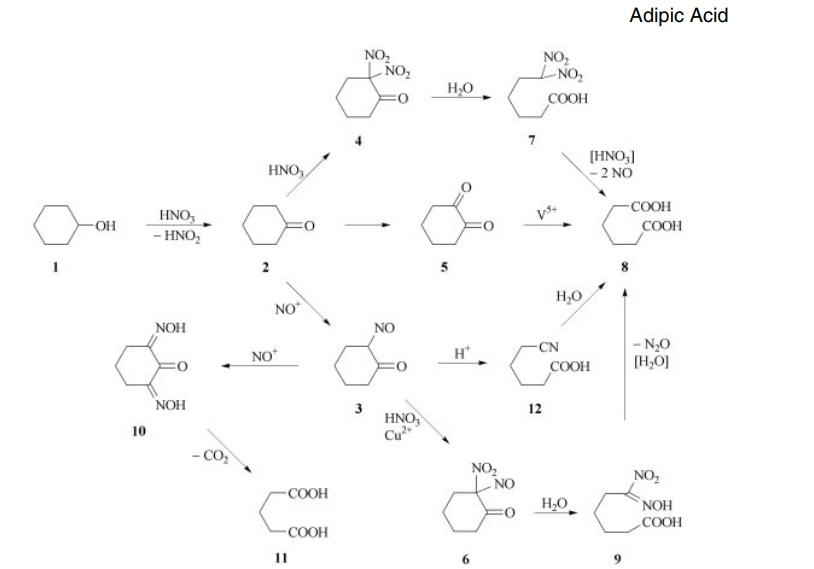
Cyclohexanol (1) oxidizes to cyclohexanone (2), along with the generation of nitrous acid. Cyclohexanone then reacts to form adipic acid (8) through one of three possible pathways. The primary route involves the nitrosation reaction to produce 2-nitrosocyclohexanone (3), which then further reacts with nitric acid to form 2-nitro-2-nitrosocyclohexanone (6). This intermediate undergoes hydrolytic decomposition to yield 6-nitro-6-hydroxyadipic acid, also known as nitro acid (9). Unrecovered nitric acid is the primary reduction product. Typically, for every mole of adipic acid produced, 2.0 moles of nitric acid are converted to nitric oxide. The second pathway occurs at higher temperatures, where the nitrosation reaction predominates. At these elevated temperatures, the pathway through dinitroketone (4) becomes significant. A third pathway proposed by early researchers involves the formation of 1,2-diketone (5) or its dimer as intermediates. To convert this material into adipic acid efficiently, vanadium catalysts are required. The impact of vanadium on overall yield suggests a significant contribution from this pathway. The intermediate nitrosocyclohexanone (3) undergoes two important side reactions. Repeated nitrosation leads to intermediate (10), which either loses carbon dioxide to yield glutaric acid (11) or subsequently reacts with nitric acid to yield succinic acid. Copper metal is added to the nitric acid to suppress these reactions. In systems with relatively high steady-state concentrations of nitrosocyclohexanone (3) or rearrangement products, Beckmann-type rearrangement leads to small amounts of 5-cyanopentanoic acid (12). This substance hydrolyzes slowly to adipic acid.
(1) Greenhouse Gases
Conventional production releases significant amounts of greenhouse gases, primarily due to the reliance on fossil fuels for energy. These emissions exacerbate global warming.
(2) Nitrogen Oxides
Nitric acid used in one step of the process releases nitrogen oxides upon reaction, contributing to smog and acid rain formation.
(3) Other Pollutants
Hydrocarbons (HC) and carbon monoxide (CO). These pollutants are released during the initial reaction stages through incomplete combustion, affecting air quality.
(4) Particulate Matter (PM)
Dust is generated during product cooling, drying, storage, and handling processes. This particulate matter can irritate the lungs, exacerbating respiratory illnesses.
(1) Water Usage
Large amounts of water are used for purification and equipment cooling, potentially straining local water resources.
(2) Process Wastewater
Wastewater generated during the production process may require treatment before discharge to avoid water pollution.
Dependency on fossil fuels. Traditional adipic acid production heavily relies on fossil fuels as energy sources, leading to greenhouse gas emissions and resource depletion.
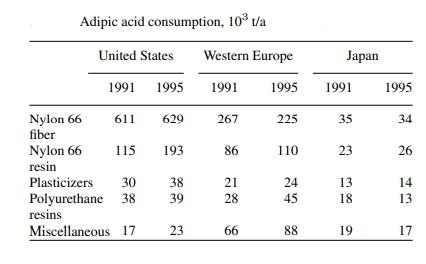
In 1999, the global annual capacity for adipic acid was 2.5 × 10^6 tons/year. While this reflects only a 15% increase in capacity since 1980, it also indicates the closure of some adiponitrile plants using adipic acid as a raw material, making it available for other purposes. North America's capacity is 1.06 × 10^6 tons/year, accounting for 42% of the total, while Western Europe accounts for 35%, mainly produced in the UK, France, Germany, and Italy. Imports and exports become significant. In 1995, the US exported 71 × 10^3 tons, accounting for 8.5% of the total US exports.
Adipic acid production is primarily driven by nylon 66 fibers and resins. Therefore, the economic outlook for this acid is largely influenced by the market dominance of these materials. Less than 15% of US production is sold in commercial markets, primarily for non-nylon uses. This proportion is higher in Western Europe and Japan. Synthesis of adiponitrile from adipic acid, once significant, is no longer used by any major manufacturer. However, in some Eastern European countries, this practice remains very limited. Non-nylon uses of adipic acid have grown at an annual rate of about 6% since 1970. Production costs are closely related to raw material prices (cyclohexane and ammonia), which declined in the late 1990s with the fall in crude oil prices. The fastest-growing regions for adipic acid and nylon 66 are China and the Far East. Growth rates in the US and Western Europe are expected to be slower, so supply and demand should remain relatively stable in the coming years.
One potential bioalternative method involves the oxidation of cyclohexanol or cyclohexanone to adipic acid by strains of Acinetobacter, Pseudomonas, and Xanthobacter bacteria. The Acinetobacter species' 14kb gene cluster for cyclohexanol oxidation contains genes encoding two alcohol dehydrogenases, an aldehyde dehydrogenase, a monooxygenase, a hydrolase, and a transcriptional regulator. Recombinant Escherichia coli containing the 14kb gene cluster can convert cyclohexanol to adipic acid. Theoretically, adipic acid could be produced from caproic acid (possibly from the Pseudomonas species), a product of Megasphaera elsdenii anaerobic glucose fermentation. This bacterium metabolizes glucose or maltose into acetic acid, butyric acid, and caproic acid through consecutive condensation of C2 units produced by pyruvic acid. Caproic acid yields of 5-8 g/l^-1 from 40 g/l^-1 glucose in batch fermentations stirred between intervals. Other potential biological processes for adipic acid production include Nocardia species producing trace amounts of acid from aliphatic amines or diamines (e.g., dodecylene diamine), and yeast converting straight-chain monocarboxylic acids (e.g., capric acid) into dicarboxylic acids, including adipic acid. Both of these biological processes are very inefficient and require significant optimization to compete with chemical processes.
This paper, by exploring methods of adipic acid preparation, demonstrates the synthetic processes and reaction mechanisms under different pathways. Through ongoing research and exploration, we can further optimize the preparation process of adipic acid, improve yield and purity, thus better meeting market demand and application requirements. It is hoped that this paper will assist readers in understanding the methods of preparing adipic acid and inspire more research and application of this compound.
[1] https://www.sciencedirect.com/science/article/abs/pii/B9780123739445001565
[2] https://onlinelibrary.wiley.com/doi/10.1002/9783527629114.ch7
[3] https://en.wikipedia.org/wiki/Adipic_acid
[4] https://www.acs.org/molecule-of-the-week/archive/a/adipic-acid.html
 |
 |
 |