D-xylose, a stereoisomer of xylose, is a common monosaccharide with significant research and application value in biochemistry, biotechnology, and other fields. Its study aids in understanding metabolic pathways in organisms and developing biotechnologies.
D-xylose, also known as D-xylose, has the chemical formula C5H10O5, with a molecular appearance of white crystalline powder or colorless needles. It serves as a crucial monosaccharide in various biochemical processes, used in food as a sweetener and preservative. Unlike other sugar alcohols, D-xylose has a lower sweetness capacity and tends to turn brown when heated. It finds application in processing industries as a sugar substitute, preservative, pet food, candies, beverages, food and animal feed, and as a reagent in cosmetics and technical applications.
Is xylose a natural sugar? Xylose, derived from the Greek word "ξ ζ λον" meaning "wood," is initially isolated from wood, hence its name. Classified as an aldo-pentose monosaccharide, it contains five carbon atoms and an aldehyde functional group. It originates from hemicellulose, one of the main components of biomass. Similar to most sugars, it can adopt several structures depending on conditions. So, is D-xylose a reducing sugar? Due to its free aldehyde group, it qualifies as a reducing sugar.
The acyclic form of xylose has the chemical formula HOCH2(CH(OH))3CHO. The cyclic hemiacetal is more common in solution, with two types: pyranose, characterized by a six-member C5O ring, and furanose, characterized by a five-member C4O ring (with a CH2OH group). Each of these rings undergoes further isomerization depending on the relative orientation of hydroxyl groups.
The dextrorotatory form, D-xylose, is a common endogenous form in organisms. It can synthesize the levorotatory form, L-xylose. The structure of D-xylose is shown in the diagram below:

D-xylose is a natural sugar found in many plant materials, with industrial and edible applications. What are the sources of D-xylose? They can be classified as follows:
D-xylose, unlike glucose or fructose, is not as abundant but exists in various plant-based foods. Here are some examples:
(1) Fruits: Varying in quantity, some fruits like berries, plums, and grapes contain small amounts of D-xylose.
(2) Vegetables: Similar to fruits, vegetables like corn, cauliflower, and green beans may contain trace amounts of D-xylose.
(3) Seeds: Seeds such as oats, rye, and flax are good sources of hemicellulose, a plant fiber containing D-xylose.
(4) Hardwoods: Birch trees, in particular, have abundant hemicellulose, serving as a natural source of D-xylose.
D-xylose, a plant-derived monosaccharide, can be obtained through fermentation on corn cob kernels. Commercial D-xylose is primarily produced by processing plant materials rich in hemicellulose. The extraction methods include:
(1) Hemicellulose Hydrolysis: This process involves breaking down hemicellulose from plant sources like corn cobs, hardwood chips, or straw using heat, acid, or enzymes. The hydrolysis releases D-xylose and other sugars.
(2) Purification: After hydrolysis, the extract undergoes purification to separate D-xylose from other sugars and impurities. Different techniques like chromatography or crystallization may be employed.
L Ricciardi et al. reported on the selective extraction of xylose from acidic hydrolysates for organic solvents, beneficial for extracting xylose into the organic phase but also allowing re-extraction into water. This process is designed around the hydrophobicity of xylose and the charge-neutral borate esters of xylose.
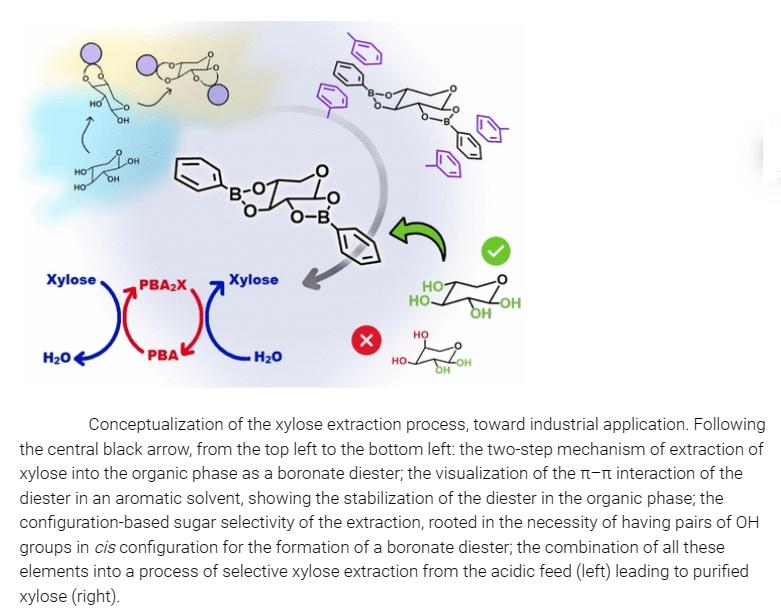
The figure illustrates the two-step mechanism of extracting xylose into the organic phase as borate esters. The visualization of π-π interactions of esters in aromatic solvents demonstrates the stability of esters in the organic phase; the configuration-based sugar-selective extraction, rooted in the necessity of pairs of OH groups to form borate esters in the cis configuration; combining all these elements into the process of selectively extracting xylose from acidic feedstocks, resulting in purified xylose.
D-xylose is absorbed in the small intestine, particularly in the upper portion called the jejunum. The jejunum is lined with millions of finger-like protrusions called villi, increasing the surface area of the small intestine, allowing for efficient absorption of nutrients from digested food.
Xylose (D-xylose) is a monosaccharide (carbohydrate) typically easily absorbed by the human body. It traverses the villi wall, enters the bloodstream, and is transported to the liver for further processing.
The D-xylose absorption test is a medical test used to measure the absorption of xylose by the small intestine. Abnormal test results may indicate issues with nutrient absorption.
D-xylose is generally considered safe. It is mainly used for:
(1) Food Additive: Many national health agencies, including the FDA, conclude that D-xylose as a food additive sweetener is safe and effective. However, excessive intake of D-xylose, like other sugar alcohols, can cause gastrointestinal discomfort and laxative effects.
(2) Medical Testing: D-xylose is also used in medical tests like the D-xylose absorption test to evaluate the absorption of nutrients by the small intestine. The D-xylose solution used in this test is generally safe but may cause some mild side effects like nausea or diarrhea.
If consumed in moderation or used for medical testing, D-xylose is generally safe for most people. If you have any concerns about D-xylose, it's best to discuss them with your doctor.
D-xylose, a stereoisomer of xylose, represents one form of this sugar. As an essential organic compound, D-xylose holds wide prospects for applications in biology, medicine, food, and other industries. Through in-depth research and development, the characteristics and functions of D-xylose can be better utilized, driving its application in various fields and bringing more benefits to human health and production.
[1]https://en.wikipedia.org/wiki/Xylose
[2]https://act.de/en/products/sweeteners-sugar-substitutes/d-xylose-wood-sugar
[3]https://pubmed.ncbi.nlm.nih.gov/1988262/
[4]https://www.testing.com/tests/xylose-absorption-test/
[5]https://pubs.acs.org/doi/pdf/10.1021/acssuschemeng.1c00167
 |
 |
 |