Dimethyl methylphosphonate (DMMP) is an important organophosphorus compound with wide-ranging applications and research value. This article aims to introduce the chemical structure, properties, and applications of DMMP across different fields. As a phosphorus-containing organic compound, DMMP plays a crucial role in industries such as chemical engineering, pesticides, pharmaceuticals, and more, owing to its various unique chemical properties and biological activities. Through the introduction and analysis of DMMP, we can better understand its significance in scientific research and industrial production, providing more information and references for related fields.
What is DMMP?
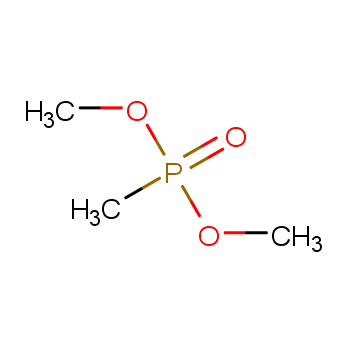
What is the full form of Dmmp? DMMP stands for Dimethyl Methylphosphonate. It is a chemical compound with the formula CH3PO(OCH3)2. Dimethyl methylphosphonate (DMMP) is a colorless liquid and a potent flame retardant used in materials like polyurethane foam, unsaturated polyester, epoxy resins, and others. DMMP is a hazardous substance and should be handled with care. It can cause irritation to the skin, eyes, and respiratory tract. Inhalation of DMMP can lead to more severe health issues such as nerve damage.
Understanding the Composition and Properties of DMMP
Exploring the Structure of DMMP
What is the structure of Dmmp? The structure of DMMP consists of:
(1) Central phosphorus atom: The phosphorus atom is the central atom in the DMMP molecule, bonded to four oxygen atoms and one methyl group. The sp3 hybridization of the phosphorus atom results in its four bonds arranged in a tetrahedral manner.
(2) Oxygen atoms: The four oxygen atoms in the DMMP molecule have different chemical environments. Two oxygen atoms bonded to the methyl group exhibit sp3 hybridization, with their three bonds arranged in a trigonal pyramid. The two oxygen atoms bonded to the carbonyl exhibit sp2 hybridization, with their three bonds arranged in a planar triangle.
(3) Methyl group: The methyl group consists of a tetrahedral structure with one carbon atom and three hydrogen atoms. The carbon atom exhibits sp3 hybridization, with its four bonds arranged in a tetrahedral manner.
Physical and Chemical Properties
What are the Uses of DMMP?
Dimethyl methylphosphonate (DMMP) is a highly versatile chemical with widespread applications across various industries. Its primary commercial use is as a flame retardant. Other commercial uses include being an early combustion additive in gasoline, a defoaming agent, plasticizer, stabilizer, textile conditioning agent, antistatic agent, as well as an additive in solvents and low-temperature hydraulic oils.
(1) DMMP in Industrial Processes: Catalysts and Intermediates
In industrial environments, DMMP serves as both a catalyst and an intermediate. It can be used as a catalyst and reagent in organic synthesis, generating highly reactive acylations. Its effective participation in chemical reactions makes it a valuable tool for manufacturing other chemicals. Additionally, DMMP can serve as a cornerstone for synthesizing more complex molecules used in various industrial applications.
(2) DMMP in Agriculture: Enhancing Crop Protection
In the agricultural sector, DMMP plays a crucial role in enhancing crop protection. Certain formulations containing DMMP can act as insecticides or fungicides, protecting crops from harmful pests and fungal diseases. This application helps ensure healthy and high-yielding agricultural output.
(3) DMMP in Chemical Warfare Agents: Understanding its Role
Among several classes of chemical warfare agents, nerve agents have been proven to be the most dangerous. Even short-term exposure to them can lead to severe toxicity. Humans can unwittingly face the aftermath of these chemicals even years after their use. Due to its much lower toxicity, dimethyl methylphosphonate (DMMP) serves as a less toxic analog of nerve agents and is frequently used as a surrogate in laboratories. DMMP has a chemical structure almost identical to nerve agents and can mimic their properties. Understanding the role of DMMP in chemical warfare is crucial. Because of its structural similarity to certain nerve agent precursors, DMMP can be used to produce these deadly chemicals. International regulations strictly control the production, trade, and storage of DMMP to prevent its use for such purposes.
Safety Precautions and Regulations
(1) Health Risks Associated with DMMP Exposure
While DMMP is beneficial in various applications, exposure to it can pose significant health risks. Inhalation, ingestion, or skin contact can cause irritation, nausea, and even nerve damage. Handling DMMP requires strict safety protocols due to its potential to disrupt the nervous system.
(2) Regulatory Guidelines and Compliance Measures
Regulatory agencies have established clear guidelines for handling and using DMMP. These guidelines typically outline requirements for personal protective equipment (PPE), proper ventilation procedures, and disposal plans. Compliance with these regulations and the implementation of appropriate measures, such as using chemical fume hoods, wearing gloves and respirators, and ensuring proper training for personnel, are crucial for reducing the risks associated with DMMP.
(3) Operations
Wear personal protective equipment/facial protection. Do not enter eyes, skin, or clothing. Use only in a chemical fume hood. Do not inhale mist/vapors/spray. Do not swallow. If swallowed, seek medical attention immediately. Keep away from open flames, hot surfaces, and ignition sources.
(4) Storage
Store containers tightly closed in a cool, dry, well-ventilated area. Keep away from heat sources, sparks, and incompatible materials, strong oxidizers.
Comparative Analysis: DMMP vs. Alternatives
(1) Advantages of DMMP over Traditional Alternatives
DMMP offers certain advantages over traditional flame retardants. Unlike some older options, DMMP typically has lower toxicity, decomposes more readily during combustion, and releases fewer harmful gases. Additionally, its compatibility with various materials makes it more effective in certain applications.
(2) Limitations and Challenges of DMMP Use
DMMP also has limitations and challenges. Concerns about its potential environmental impact arise because some studies suggest it may decompose into harmful byproducts. Furthermore, the aforementioned role of DMMP as a precursor to nerve agents requires strict regulation, which may affect its availability and increase its cost.
Conclusion
While the versatility of dimethyl methylphosphonate (DMMP) brings undeniable benefits across industries, its potential health risks and environmental concerns require responsible handling and ongoing research. By implementing stricter regulations, exploring green alternatives for specific applications, and continuously improving safety protocols, we can harness the potential of DMMP while mitigating its associated risks.
References:
[1]https://www.ashland.com/file_source/Ashland/Documents/Sustainability/rc_dimethyl_methylphosphonate_pss.pdf
[2] https://pubmed.ncbi.nlm.nih.gov/38394750
[3] https://en.wikipedia.org/wiki/Dimethyl_methylphosphonate
[4] https://www.fishersci.com>