Dioctyl adipate (DOA) serves as a crucial organic compound with significant roles across various industries. This article aims to provide a brief overview of DOA, including its chemical structure, properties, and applications in different sectors. As a type of diester compound, DOA possesses unique chemical properties and diverse uses. By delving into the importance of DOA in various industries, we can gain a better understanding of its wide-ranging application value, providing more information and guidance for research and production in related fields.
Introduction to Dioctyl Adipate (DOA)
Plasticizers are widely used additives in plastic products. They enhance the plasticity of polymers, improve processability, and impart flexibility to products. Plasticizers constitute one of the largest categories of chemicals in terms of production and consumption, accounting for about 60% of the total output of plastic additives. In recent years, plastic products have been evolving towards lightweight, composite, and functional directions, posing new demands on plasticizers. Manufacturers and research institutions need to continuously develop new products and adjust product structures to meet the evolving needs of the plastic industry.
Dioctyl adipate (DOA) is a cold-resistant plasticizer for polyvinyl chloride (PVC), possessing certain heat, light, and water resistance. It is commonly used in the processing of plastics, rubber products, and synthetic resins as a plasticizer. DOA is often used in combination with primary plasticizers such as DOP for cold-resistant agricultural films, wires, thin sheets, synthetic leather, and packaging films for frozen foods. It can also serve as a low-temperature plasticizer for many synthetic rubbers and resins such as nitrocellulose.
What is Dioctyl Adipate (DOA) and What Solvent Does it Use? What is the solvent for dioctyl adipate?
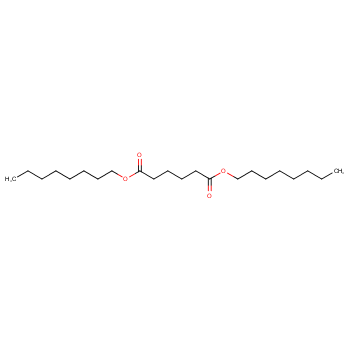
Dioctyl adipate (DOA) is an organic compound belonging to the ester class, with a chemical formula of (CH2CH2CO2C8H17)2. Its molecular structure consists of two octyl groups (C8H17) connected by a central adipate group (CH2CH2CO2). DOA is a non-polar molecule, meaning it tends to dissolve well in other non-polar solvents. Some examples of good solvents for DOA include aliphatic hydrocarbons such as hexane and heptane. The structure of dioctyl adipate is as follows:
This structure imparts several key properties to dioctyl adipate (DOA):
● (1) Low-temperature flexibility: DOA maintains flexibility even at low temperatures, ensuring optimal performance in cold environments.
● (2) Electrical performance: DOA enhances the electrical properties of materials, making it suitable for applications in the electrical and electronics industries.
● (3) Chemical resistance: DOA exhibits excellent resistance to oil, grease, and chemicals, ensuring durability and longevity in harsh environments.
● (4) Reduction in glass transition temperature: DOA effectively lowers the glass transition temperature of polymers, improving their flexibility and processing performance.
Applications of Dioctyl Adipate (DOA)
● (1) Flexible Films and Sheets: Widely used in the production of flexible films and sheets, making it an ideal choice for packaging materials and protective coverings.
● (2) Cables and Wires: Utilized for insulation and sheathing of cables and wires, providing flexibility and oil resistance to ensure reliable electrical performance.
● (3) Hoses and Pipes: Enhances the flexibility and chemical resistance of hoses and pipes, making them suitable for applications in automotive, construction, and manufacturing industries.
● (4) Coated Fabrics: Acts as a plasticizer in coated fabrics such as synthetic leather and textiles, improving their flexibility, durability, and oil resistance. This makes them ideal choices for interior decoration, automotive interiors, and footwear.
● (5) Other Applications: DOA can also be used in various other applications including gaskets, seals, adhesives, and printing inks, where its plasticizing and performance-enhancing properties are highly valuable.
Safety Precautions: Is dioctyl adipate hazardous?
Dioctyl Adipate has low toxicity, with an oral LD50 of 3000 to 6000 mg/kg in rats, and minimal irritation to skin and eyes. While DOA is considered to be non-toxic, it is important to handle it with care to avoid potential hazards. Regulatory bodies like OSHA (Occupational Safety and Health Administration) have not established specific exposure limits, but Safety Data Sheets (SDS) typically recommend precautions against skin and eye irritation. Therefore, safe handling practices include wearing gloves, safety goggles, and working in well-ventilated areas to minimize contact and inhalation. Additionally, DOA is flammable, so keeping it away from heat sources and storing it properly is crucial for fire safety.
Dioctyl adipate synthesis
How is dioctyl adipate made? Traditional synthesis of DOA typically involves adipic acid and 2-ethylhexanol as raw materials, catalyzed by concentrated sulfuric acid to undergo esterification. However, the process requires purification steps such as activated carbon decolorization, neutralization, water washing, and alcohol removal to obtain the final product. This method suffers from product losses, equipment corrosion, and generates a considerable amount of waste residue and water pollution. Given the various drawbacks of sulfuric acid catalysis, developing new, high-quality, efficient, low-corrosion, easily separable, reusable, and cost-effective catalysts has become an important task in DOA synthesis, aligning with the current trend in catalyst development.
(1) Organic Acids
Li Nan et al. utilized microwave radiation technology with toluenesulfonic acid supported on activated carbon as a catalyst to directly esterify adipic acid and n-octanol to synthesize DOA. Through orthogonal experiments, the optimal reaction conditions were determined as follows: adipic acid 1 mmol, n-octanol 4 mmol, catalyst dosage 0.8 g, microwave power 600 W, radiation time 45 s, achieving a conversion rate of 99.3%. The catalyst could be reused.
(2) Solid Superacid
Zhang Yunhuai used rare earth composite solid superacid SO2-4/ZrO2-La2O3 as a catalyst for DOA synthesis. The optimal process conditions were determined as follows: catalyst dosage 5%, alcohol-to-acid molar ratio 2.8:1, reaction temperature 130-140°C, reaction time 3 hours, achieving an esterification rate of 97.6%. The catalyst could be reused multiple times with consistent activity even after 6 repetitions, thanks to the addition of rare earth element La, which enhanced the catalytic performance and prolonged the lifespan of the solid superacid.
(3) Activated Carbon Fiber
Meng Qi et al. synthesized DOA catalyzed by SnO-loaded activated carbon fiber. The optimal process conditions were: n(adipic acid):n(2-ethylhexanol) = 1:3.3, catalyst 1.1%, toluene as the water-carrying agent at 6%, reaction temperature 170-175°C, reaction time 120 minutes, achieving an esterification rate of 98.2%. This method allowed obtaining qualified products without the need for neutralization, water washing, and decolorization, thus achieving a cleaner production process compared to traditional methods.
(4) Heteropolyacid
Luan Xianghai et al. utilized environmentally friendly phosphotungstic heteropolyacid as a catalyst, with adipic acid and n-octanol as raw materials, and toluene as the water-carrying agent, to synthesize dioctyl adipate. An apparent kinetic model for esterification synthesis was established. Through uniform design experiments, the optimal process conditions were determined as follows: alcohol-to-acid molar ratio of 2.8:1, catalyst dosage of 1.2% of acid mass, water-carrying agent dosage of 75% of acid mass, reaction time 3 hours, achieving an esterification rate of 99.23%.
Environmental Impact and Sustainability
6.1 Environmental Hazards: Classified as acute hazards to aquatic environments, category 1; and long-term hazards to aquatic environments, category 1.
6.2 Environmental Precautions: Prevent further leakage or spillage under safe conditions. Avoid water contamination. Refrain from releasing into the environment. Manage and treat contaminated wash water. In case of spills into drainage/aquatic environments, contact local authorities.
6.3 Sustainability: Compared to other plasticizers, DOA offers favorable environmental benefits, primarily its biodegradability. While its production still consumes resources and energy, DOA's advantages include rapid biodegradation, minimizing its long-term presence in the environment. However, researchers continually seek more sustainable solutions, such as bio-based plasticizers extracted from renewable resources like plant oils, to further reduce the environmental footprint of these materials.
Conclusion: Embracing the Potential of Dioctyl Adipate (DOA)
DOA stands as a multifunctional and valuable chemical. Its colorless oily liquid nature encompasses numerous properties like low-temperature flexibility, electrical stability, and weather resistance, making it a preferred plasticizer for various applications. While safety precautions are necessary during handling, DOA's biodegradability sets it apart from some alternatives. With ongoing research into sustainable practices, bio-based plasticizers offer exciting prospects for the future. The current advantages of DOA and ongoing exploration of its diversified applications consolidate its role as a significant contributor across various industries.
References:
[1] Fu Junhong, Preparation of high-purity dioctyl adipate (DOA). Zhejiang Province, Zhejiang Jiaao Environmental Protection Technology Co., Ltd., 2021-05-28.
[2] Chen Tianzhi. Preparation and catalytic synthesis of dioctyl adipate using carbon sulfonic acid from lignin and polyphenylene ether-based resin [D]. Hunan Normal University, 2016.
[3] Yin Xiuyun, Li Gan. Research on the synthesis of dioctyl adipate [J]. Guangzhou Chemical Industry, 2014, 42 (13): 24-26.
[4]https://www.parchem.com/siteimages/attachment/ghs%20dioctyl%20adipate%20msds.pdf
[5]https://www.pishrochem.com/blog/en/doa-dioctyl-adipate-properties-applications-and-benefits/