-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

Ethyl cinnamate, also known as β-phenylacrylic acid ethyl ester, is an important fragrance and food additive, with a scent reminiscent of sweet oranges and grapes, lasting and profound. It is also used in cigarette tobacco as a flavoring and aroma compensator. In addition, ethyl cinnamate also exhibits certain biological activities such as antibacterial, anti-inflammatory, and antitumor effects. The demand for ethyl cinnamate is increasing year by year, and traditional synthesis methods have some drawbacks, such as harsh reaction conditions, catalyst environmental pollution, low yield, etc. Therefore, the development of new, efficient, and environmentally friendly ethyl cinnamate synthesis methods for ethyl cinnamate is of great theoretical value and practical significance.
Ethyl cinnamate naturally occurs in storax (resin secreted by the Liquidambar orientalis) and ylang-ylang flowers, with its molecular structure shown in Figure 1.
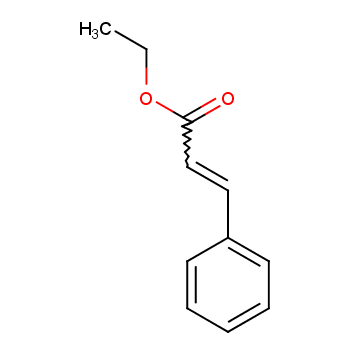
Currently, ethyl cinnamate is widely used in the food, cosmetics, and pharmaceutical fields, among others. It plays an important role in various industries. Due to economic, productivity, and environmental reasons, there is an urgent need for efficient preparation methods, considering factors such as catalysts, desiccants, reaction time, and raw material usage. Traditionally, ethyl cinnamate is synthesized by the esterification of cinnamic acid with ethanol under sulfuric acid catalysis. However, this method suffers from long reaction times, poor selectivity, multiple side reactions, low yield (about 60%), severe equipment corrosion by sulfuric acid, difficult post-treatment of products, and serious pollution. With the increasing environmental awareness, finding new high-yield synthetic methods has become a new research topic.
Ethyl cinnamate, a versatile aromatic compound, can be synthesized through three main methods:
This method involves the reaction of cinnamic acid (C6H5CH=CHCOOH) with ethanol (C2H5OH) in the presence of an acid catalyst (usually sulfuric acid (H2SO4)). In the presence of hydrogen chloride or sulfuric acid, ethanol reacts with cinnamic acid, while in the presence of sodium, ethyl acetate reacts with benzaldehyde to yield ethyl cinnamate.
To understand the basic principle of the Claisen-Schmidt reaction, a detailed exploration of its mechanism is required. This is a two-step process initiated by ester deprotonation.
Step 1: The initial step involves ester deprotonation, leading to the formation of an enolate ion. A strong base used in the reaction extracts a proton from the ester's α-carbon, producing this crucial enolate ion.
Step 2: In the second step, this enolate ion acts as a nucleophilic reagent and attacks the carbonyl carbon of the second ester molecule. This process results in the elimination of the alkoxy group and the formation of a new C-C bond, thereby forming β-keto esters or β-diketones.
The ethyl cinnamate synthesis reaction involves the condensation of benzaldehyde (C6H5CHO) and ethyl acetate (CH3COOC2H5) in the presence of a strong base catalyst (such as sodium ethoxide (NaOC2H5)). The base removes a proton from the α-carbon of ethyl acetate, forming a nucleophilic reagent that attacks the carbonyl carbon of benzaldehyde. This forms a new carbon-carbon bond, eliminating ethanol and yielding ethyl cinnamate.
Yun Wang et al. studied the enzymatic ethyl cinnamate synthesis by lipid enzyme TLIM catalyzing the esterification of cinnamic acid with ethanol. In order to improve the yield of ethyl cinnamate, several media were studied in this reaction, including acetone, iso-octane, DMSO, and solvent-free media. Using iso-octane as the reaction medium, the reaction showed a high yield, with a significantly higher yield than previously reported. In addition, parameters such as oscillation rate, water activity, reaction temperature, substrate molar ratio, and enzyme loading have significant effects on the reaction. For example, when the temperature increased from 10°C to 50°C, the initial reaction rate increased by 18 times, and the yield of ethyl cinnamate increased by 6.2 times. Under optimal conditions, the maximum yield of ethyl cinnamate catalyzed by lipase was 99%, which has universal significance for the development of industrial processes for preparing ethyl cinnamate.
Using phosphotungstic acid as a catalyst, ethyl cinnamate is synthesized by dehydration. The experiment showed that when the molar ratio of alcohol to acid substance was 13:1, the catalyst dosage accounted for 2.7% of the total mass of the reactants, and the desiccant dosage accounted for 27% of the total mass of the reactants, the reaction time was 3 hours, and the ester yield reached a maximum of 84.0%.

The specific steps are as follows: Add 3.0 g (0.02 mol) of cinnamic acid and a certain amount of phosphotungstic acid catalyst and anhydrous ethanol to a 50 mL round-bottom flask. Add a certain amount of desiccant (anhydrous magnesium sulfate wrapped in filter paper) to the constant-pressure dropping funnel. Heat reflux for a period of time, then convert to a distillation apparatus to distill off the unreacted ethanol in the flask (which can be recycled). After cooling, add 10 mL of water to the flask and extract with 40 mL of ether three times. The ether layer is washed with saturated sodium carbonate solution to remove unreacted cinnamic acid, and then separated. After drying the ether layer, evaporate the ether (recoverable) in a water bath, and cool the remaining liquid in the flask to obtain the product.
Using cinnamic acid and ethanol as raw materials, ethyl cinnamate is synthesized using anhydrous calcium sulfate as a catalyst. The optimal process conditions are: n(cinnamic acid): n(ethanol) = 1:6.36, calcium sulfate addition amount 12.86%, reaction time 14.55 hours, and ethyl cinnamate yield can reach 89.42%. It has low cost and is suitable for industrial production.
The specific steps are as follows: Add cinnamic acid, anhydrous ethanol, and anhydrous calcium sulfate to a four-neck flask equipped with a condenser and a separator, stir, heat to reflux, and continuously add water remover to the flask. The water generated by the reaction is separated out through the separator. After the reaction is completed, cool to room temperature, filter to remove the catalyst, and distill off the remaining ethanol and water under reduced pressure to obtain ethyl cinnamate. Yield: 89.4%, purity: 99.7%, melting point: 6.7~7.9°C, 1HNMR (DMSO-d6, 400 MHz), δ: 1.32 (t, 3H, -CH3), 4.25 (q, 2H, -CH2), 6.39 (d, 1H, α-HC=), 7.61 (d, 1H, β-HC=), 7.71 (br, 3H, 3,4,5-H), 7.52 (br, 2H, 2,6-H).
Prepare a microemulsion by mixing an ionic liquid with water, and then prepare nano-scale palladium catalysts by dropwise addition of palladium chloride solution in the microemulsion. Use this palladium catalyst system as the reaction medium, add iodobenzene, ethyl acrylate, and triethylamine to the system for a certain period of time to undergo a Heck reaction, thereby preparing ethyl cinnamate. This method has higher reaction efficiency for preparing ethyl cinnamate and the catalyst can be reused multiple times. Meanwhile, the yield of ethyl cinnamate exceeds 99%, demonstrating its high promotional value.
The specific experimental steps are as follows:
(1) Add 120g of [BMIM]PF6 ionic liquid to the reaction bottle, and stir magnetically at 25°C for 15 minutes.
(2) Slowly add 160g of Triton X-100 type emulsifier to the reaction bottle equipped with a condenser and a separator, while continuously bubbling nitrogen into the system to dissolve the two in each other under stirring conditions.
(3) Slowly add distilled water to the reaction bottle at a rate of 2ml/min until the entire reaction system becomes transparent and clear. Then, heat to 50°C, and when the temperature stabilizes, add 70g of palladium chloride solution to the reaction bottle and continue stirring.
(4) When the system changes from light yellow to dark black, and there is no precipitate in the reaction bottle and the system is uniform, slowly raise the temperature to 80°C, add 200g of iodobenzene, 210g of ethyl acrylate, and 210g of triethylamine to the system, and stir for 3 hours.
(5) After the reaction is completed, separate the product, confirm the product as ethyl cinnamate by nuclear magnetic resonance analysis, and calculate the yield, which reaches 99.5%.
Various techniques are used to analyze the quality and purity of ethyl cinnamate. Here are some commonly used methods:
Infrared (IR) Spectroscopy: This technique identifies functional groups present in the molecule, confirming the presence of ester and aromatic functional groups specific to ethyl cinnamate.
Ultraviolet (UV) Spectroscopy: This method can determine whether conjugated double bonds are present in the molecule, further supporting the identification of ethyl cinnamate.
High-Performance Liquid Chromatography (HPLC): This technique separates components based on polarity. By comparing the retention time of the ethyl cinnamate peak with standards and using calibration methods, the purity of the sample can be determined.
Gas Chromatography-Mass Spectrometry (GC-MS): This combination technique separates components based on volatility and then identifies and quantifies them by mass spectrometry. GC-MS can be used to identify and quantify impurities in ethyl cinnamate samples.
Nuclear Magnetic Resonance (NMR) Spectroscopy: This method provides detailed information about the molecular structure by identifying specific chemical environments of protons and carbons. NMR is used to confirm the structure of ethyl cinnamate and identify any unknown impurities.
Maintaining quality throughout the synthesis process is crucial. Here are some key aspects of quality control:
Ethyl cinnamate must comply with regulations set by organizations such as the Food and Drug Administration (FDA) or the European Pharmacopoeia (Ph. Eur.) depending on its intended use (e.g., food flavoring, pharmaceutical ingredient). These regulations specify purity requirements, acceptable levels of impurities, and any restrictions on solvents or additives used in ethyl cinnamate synthesis.
Manufacturers establish testing protocols throughout the production process. These protocols may include:
Analysis of starting materials to ensure the purity of precursors used in ethyl cinnamate synthesis.
Process controls to monitor reaction progress and identify any deviations.
Final product testing using the aforementioned analysis methods (HPLC, GC-MS, etc.) to confirm purity, characteristics, and absence of harmful contaminants.
By adhering to strict quality control measures, manufacturers can ensure continuous production of high-purity and safe ethyl cinnamate for its intended applications.
Ethyl cinnamate is an important fragrance and food additive with wide-ranging applications. Currently, ethyl cinnamate synthesis methods for ethyl cinnamate mainly include esterification and biological methods. Esterification is the main method used in industrial production of ethyl cinnamate, but it suffers from drawbacks such as catalyst pollution of the environment. Biological methods are environmentally friendly, but they have slower reaction rates and higher costs. With the advancement of science and technology, new ethyl cinnamate synthesis methods for ethyl cinnamate are expected to emerge continuously. In the future, ethyl cinnamate synthesis methods for ethyl cinnamate will develop towards efficiency, environmental friendliness, and low cost.
[1] Cheng, D., & Feng, M. (2024). Integration of Project-Based Teaching with Professional Orientation - Industrial Synthesis of Ethyl Cinnamate. Chemistry Education (Chinese/English), 45(01), 40-46. DOI:10.13884/j.1003-3807hxjy.2022100182.
[2] Hao, L., & Zhang, L. (2019). Research on the Synthesis of Ethyl Cinnamate Catalyzed by Calcium Sulfate. Contemporary Chemical Industry, 48(10), 2223-2226. DOI:10.13840/j.cnki.cn21-1457/tq.2019.10.012.
[3] Liu, K. (2016). Synthesis of Ethyl Cinnamate by Dehydration Method Catalyzed by Phosphotungstic Acid. Guangdong Chemical Industry, 43(05), 95-96+92.
[4]https://www.sciencedirect.com/science/article/abs/pii/S030881461500895
[5]https://www.vaia.com/en-us/explanations/chemistry/organic-chemistry/claisen-condensation/
 |
 |
 |