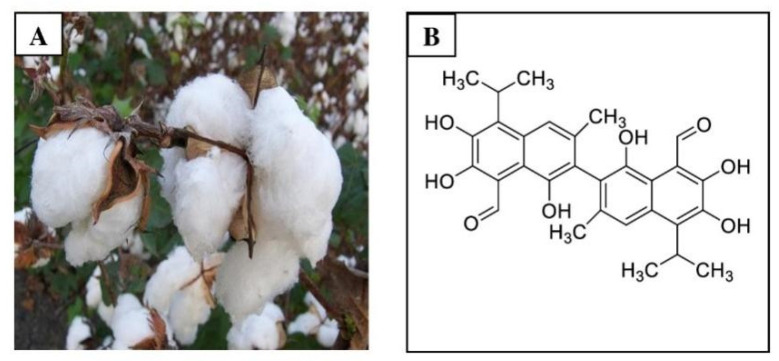
Discovered by Langmuir in 1886 and later purified by Marchlewski in 1899, gossypol is derived from cotton plants and characterized by its polyphenolic nature. It exhibits various biological activities, including contraceptive effects originally explored in male contraception. Notably, gossypol's chemical composition includes polyphenolic properties, making it a subject of numerous studies.
Gossypol is a bright yellow pigment found in the glandular cells of cotton stems, leaves, seeds, root bark, cotton petals, seed coats, husks, and flowers. Factors influencing gossypol levels include fertilizer usage, composition, cotton variety, planting time, regional temperature, soil conditions, moisture distribution, and agricultural techniques. Plants containing gossypol include:

Applications:
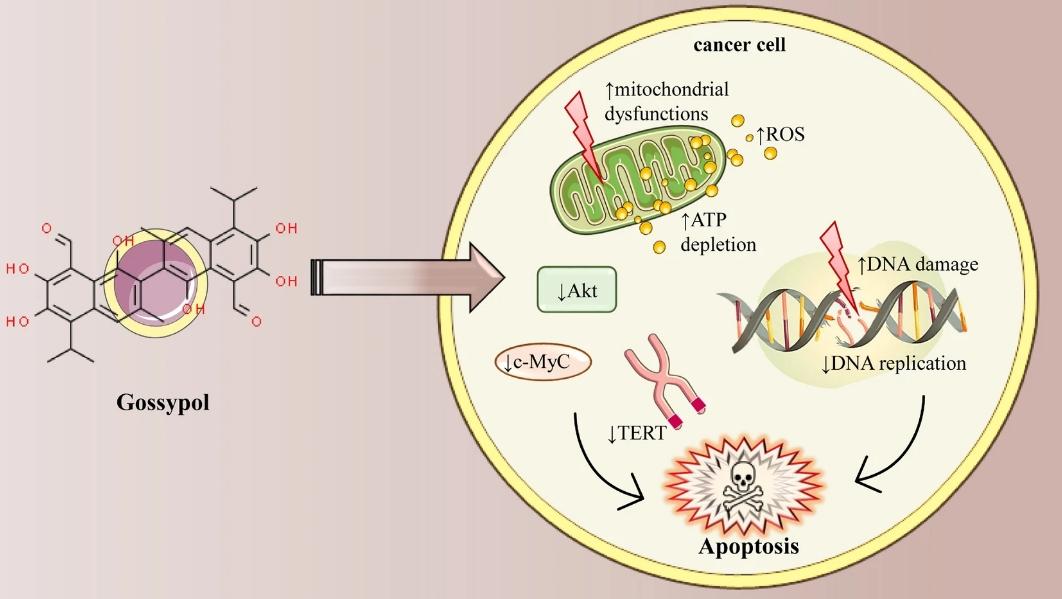
Despite decades of research and significant findings, cancer remains a leading cause of global mortality and a major public health issue. Gossypol, a natural polyphenolic compound found in cotton (Gossypium hirsutum L.) seeds, roots, and stems, initially served as a male contraceptive. Due to its biological properties such as antifertility, antiviral, antioxidant, antibacterial, antimalarial, and notably, anticancer activity, gossypol has become the focus of extensive research.
Gossypol Side Effects:
Gossypol is toxic, contributing to protecting cotton plants from various insects and/or pathogens. Consequently, animal feed cottonseed meal may exhibit long-term toxicity. Moreover, it can directly or indirectly become a source of human toxicity through the food chain. Free gossypol exposure may lead to anorexia, respiratory distress, impaired weight gain, indifference, compromised immunity, cellular and vascular damage, heart failure, and potential fatality. Major toxicities include male infertility, which may be irreversible, and hypokalemia. Clinical studies have identified gossypol's significant side effects, including hemolytic anemia, diarrhea, and other gastrointestinal symptoms. Various detoxification methods for cottonseed meal include dry heating, soaking, puffing, and centrifugal separation. Chemical detoxification involves extraction, oxidant oxidation, and alkali soaking. Furthermore, microbial fermentation can reduce free gossypol toxicity. Additionally, managing significant gossypol side effects may involve dose reduction and symptomatic treatment as feasible. Derivatization of gossypol could potentially enhance biological potency and reduce toxicity.
Common Gossypol Side Effects:
(1) Oral Consumption:
Daily gossypol doses not exceeding 20 mg, taken continuously for one year, may be safe. However, excessive doses or prolonged use beyond one year may pose safety risks. Overdosing may lead to malnutrition, gastric bleeding, heart failure, and liver issues. Excessive doses and prolonged use are also more likely to result in permanent infertility.
(2) Topical Application:
Currently, there is insufficient reliable information to determine the safety or potential side effects of topical gossypol application.
Clinical Studies on Gossypol Adverse Reactions:
The irreversible impact of gossypol on male fertility and the incidence of hypokalemia have been well documented. Clinical trials assessing gossypol in cancer applications have reported adverse effects such as nausea, vomiting, anorexia, diarrhea, taste alterations, intestinal obstruction, and fatigue. Limitations in liver enzymes have been noted with a total gossypol dose of 60 mg/day. While no clinically significant hematologic toxicity was observed when gossypol was used alone in cancer research, additional marrow suppression appears when combined with chemotherapy drugs, varying by regimen. In a phase 1 trial, hematologic toxicity increased with 40 mg of gossypol, mitigated when non-gestational age was added to the cisplatin-etoposide treatment regimen. Schleman 2014.
In a 2016 scientific statement from the American Heart Association on drugs that may cause or exacerbate heart failure, gossypol was considered a product with potentially harmful effects on the cardiovascular system (e.g., enhancing the action of diuretics), which may harm heart failure patients. The statement advised against using nutritional supplements for managing heart failure symptoms or for secondary prevention of cardiovascular events.
Precautions:
(1) Pregnancy:
Oral gossypol during pregnancy may be unsafe and could lead to miscarriage. Avoid use.
(2) Breastfeeding:
Oral gossypol during breastfeeding may be unsafe. Use caution and avoid.
(3) Hypokalemia:
Gossypol may worsen hypokalemia. Avoid use if you have this condition.
Managing and Mitigating Side Effects:
Safety Guidelines:
(1) Adhere to Recommended Dosages:
Always strictly follow dosage instructions. Higher doses are more likely to cause side effects such as malnutrition, gastric issues, and heart problems.
(2) Limit Prolonged Use:
If using gossypol products long-term, consult healthcare professionals to monitor potential issues.
(3) Maintain a Balanced Diet:
Gossypol can interfere with nutrient absorption. Ensure you get adequate essential vitamins and minerals through a healthy diet and consider supplements when necessary.
Personal Tips for Using Gossypol Products:
(1) Watch for Interactions:
Gossypol may interact with certain medications. Before using gossypol products, discuss any medications you are taking with your doctor.
(2) Monitor Your Body:
Pay attention to how you feel when using gossypol. If you experience any concerning side effects, discontinue use and consult healthcare professionals.
(3) Consider Alternatives:
If managing side effects becomes challenging, explore alternatives to achieve your desired goals.
Future Research:
Ingesting gossypol from cottonseeds and their products (cakes and flours) may promote clinical poisoning, liver damage, male and female reproductive toxicity, and immune dysfunction. Acute poisoning is currently not a significant issue, but reproductive damage poses serious economic losses to livestock. While male reproductive toxicity is well-documented, more research is needed to understand gossypol-induced female reproductive damage. The immunotoxicity of gossypol remains incompletely elucidated but affects animals by reducing resistance to infection and impairing vaccine efficacy. Extensive research is needed to develop more effective and affordable technologies to reduce gossypol toxicity.
Conclusion:
While gossypol holds potential in medicine, its gossypol side effects cannot be overlooked. When using gossypol, it is crucial to consult healthcare professionals to ensure safety and effectiveness. Only through comprehensive assessment and proper guidance can we fully harness its potential while minimizing possible adverse effects.
References:
[1]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4033412/
[2]https://cmjournal.biomedcentral.com/articles/10.1186/s13020-023-00869-8
[3]https://www.drugs.com/npp/gossypol.html
[4]https://www.webmd.com/vitamins/ai/ingredientmono-106/gossypol
[5]https://today.tamu.edu/2019/10/14/fda-approves-ultra-low-gossypol-cottonseed-for-human-animal-consumption/
[6]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9787675/