"Is guanidine nitrate toxic?" has long been a focal point of concern and discussion. Through a comprehensive study of its toxicological characteristics, a more thorough evaluation of the potential risks and impacts of its practical applications can be achieved.
Is guanidine nitrate toxic? Guanidine nitrate, with the chemical formula CHN6O3, is an organic compound. It is a white, odorless crystalline solid, soluble in water and other polar solvents. Guanidine nitrate is utilized as a fertilizer, propellant, and gas generator in automotive safety airbags. Upon heating, guanidine nitrate decomposes, producing nitrogen gas and various other by-products. This decomposition reaction is rapid and exothermic, making guanidine nitrate a useful propellant and gas generator.
In automotive safety airbags, guanidine nitrate is employed to generate nitrogen gas for inflating the airbags. Upon collision, sensors trigger the release of guanidine nitrate from the inflator. Subsequently, guanidine nitrate rapidly decomposes, generating a large volume of nitrogen gas to inflate the safety airbag.
Guanidine nitrate may explosively decompose upon impact, friction, or shock. It is prone to explosion when heated, producing toxic, corrosive smoke containing nitric acid and nitrogen oxides during combustion. This substance acts as a strong oxidizing agent and reacts with combustible and reducing materials.
Guanidine nitrate exhibits "slight" toxicity to mice, with an oral LD50 of approximately 1,100 mg/kg (Brown et al., 1988). Other sources listed in the Hazardous Substances Data Bank (HSDB) (NLM, 2008) report slightly different values for rats (989.6 mg/kg [males], 729.8 mg/kg [females]), with males mice at 1,105 mg/kg and females at 1,029 mg/kg (gavage administration). The acute inhalation toxicity in rats (LC50) (4 hours duration) is reported to be >0.853 mg/L (>0.853 mg/m3) (Brown et al., 1988). Skin exposure in rabbits at 2 g/kg produced only mild skin irritation in the same report.
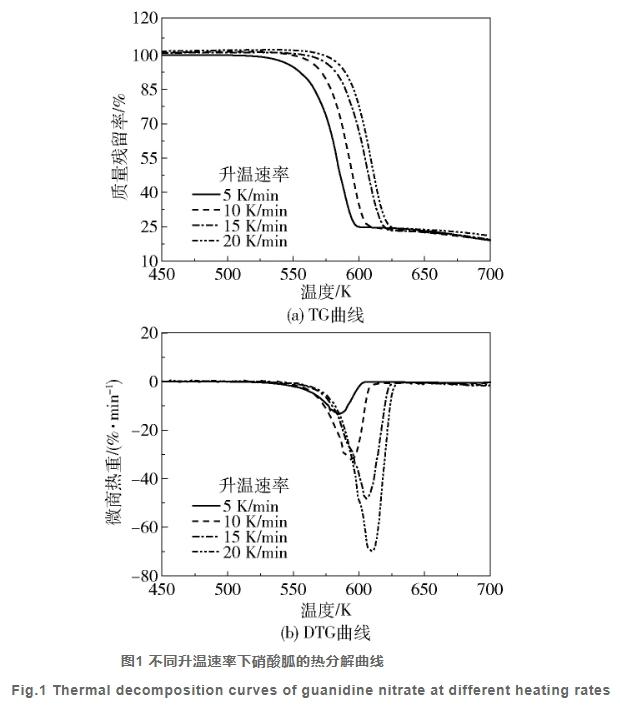
Due to its strong oxidizing nature, guanidine nitrate may explosively decompose when exposed to heat, flame, friction, vibration, or impact. When heated to 150°C, guanidine nitrate decomposes and explodes. The direct cause of the Krell Company explosion accident was a leaking discharge valve (heated with thermal oil) at the bottom of reactor No. 1, which ignited the thermal oil, causing localized heating of guanidine nitrate and unreacted ammonium nitrate, resulting in rapid decomposition and explosion, triggering explosions of stored guanidine nitrate and ammonium nitrate. Mixtures of nitro compounds and chlorates also explode upon vibration and friction. Toxic nitrogen oxides and other combustion products are released during high-temperature decomposition.

Contact with other substances may cause fires. It may lead to severe eye, skin, and respiratory irritation, and potential burns. Ingestion may be harmful.
(1) Eyes: May cause conjunctivitis. May lead to permanent corneal opacification.
(2) Skin: May cause severe irritation and possible burns.
(3) Ingestion: May lead to gastrointestinal burns. Ingestion of compounds containing nitrates can cause methemoglobinemia. May result in nausea, vomiting, and diarrhea, possibly with blood.
(4) Inhalation: May cause acute pulmonary edema, asphyxia, chemical pneumonitis, and upper respiratory tract obstruction caused by edema.
Concerns arise regarding the environmental impact of guanidine nitrate. Limited studies on its persistence and bioaccumulation exist, yet its high water solubility suggests easy dispersion in aquatic environments. Furthermore, documented toxicity towards aquatic organisms exists. Conversely, research indicates potential biodegradation under certain conditions. Sustainable prospects remain unclear. While its explosiveness demands careful handling in production and disposal processes, studies suggest wastewater treatment can undergo bioremediation, offering a more sustainable solution. Further data is needed to precisely assess guanidine nitrate's overall environmental impact and the true sustainability of its lifecycle.
Guanidine nitrate exhibits irritant effects on eyes, skin, mucous membranes, and the upper respiratory tract, with excessive inhalation potentially leading to fatality. Nitrogen oxide gases released at high temperatures exert strong irritant effects on the respiratory tract and eyes.
Explosions of guanidine nitrate may cause severe harm to humans. The dense smoke and fog generated post-explosion induce intense irritation in the eyes and upper respiratory tract. Survivors report rapid onset of eye pain and near-instantaneous blindness, with journalists experiencing respiratory distress upon visiting the accident site the following day.
(1) Eyes: Immediately flush eyes with copious amounts of water for at least 15 minutes, occasionally lifting eyelids. Seek medical attention.
(2) Skin: Seek medical attention. Remove contaminated clothing and shoes, and flush skin with plenty of water for at least 15 minutes. Launder clothing before reuse.
(3) Ingestion: Do not induce vomiting in unconscious individuals. Seek medical attention. If conscious, rinse mouth and drink 2-4 cups of milk or water.
(4) Inhalation: Immediately evacuate the exposed area and move to fresh air. Administer artificial respiration if breathing stops. Oxygen therapy in case of respiratory distress. Seek medical attention. Do not perform mouth-to-mouth resuscitation.
(5) Notes to Physician: Symptomatic and supportive treatment.
Wear self-contained breathing apparatus, MSHA/NIOSH (approved or equivalent), and full protective gear, as with any fire. Strong oxidizer. Contact with other substances may cause fire. Thermal decomposition or combustion during fire may produce irritating and toxic gases. Handle water cautiously and use abundantly. Some oxidizers may react explosively with hydrocarbons (fuels), accelerating combustion. Containers may explode when heated.
Immediately contact professional firefighters. For small fires, do not use dry chemical, carbon dioxide, halogenated hydrocarbons, or foam. Water only. For large fires, flood fire area from a distance with water.
Due to the hazardous nature of guanidine nitrate, operations should involve: enclosed handling, providing adequate local exhaust ventilation; personnel must undergo specialized training and strictly adhere to operational procedures.
Operational personnel are advised to take the following measures: wear hood-type powered air-purifying respirators, don chemical-resistant suits, and neoprene gloves. Keep away from sources of heat and fire, strictly prohibit smoking, maintain workplace cleanliness. Avoid contact with flammable materials, and minimize dust generation. Handle with care during transport to prevent damage to packaging or containers. Avoid vibration, impact, and friction, ensuring operational safety. Additionally, equip with necessary firefighting equipment and spill containment devices. Even empty containers may contain hazardous substances; personnel must handle with care.
Store in a cool, ventilated warehouse. Keep away from ignition sources and heat. Seal packaging. Do not store with strong reducing agents, flammable or combustible materials, sulfur, phosphorus, etc., to prevent mixing. Storage areas should be equipped with appropriate materials for containing spills.
(1) Eyes: Wear appropriate protective goggles or chemical safety goggles as described in OSHA's eye and face protection standard 29 CFR 1910.133 or European standard EN166.
(2) Skin: Wear suitable protective gloves to prevent skin exposure.
(3) Clothing: Wear chemical aprons. Wear appropriate protective clothing to prevent skin exposure.
(4) Respirator: Respirators must be used where workplace conditions allow, following OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European standard EN 149 respiratory protection program.
In conclusion, continuous in-depth research and assessment are necessary regarding the toxicity of guanidine nitrate. While it may present toxicity in certain scenarios, proper usage and management can minimize its potential hazards. Through ongoing scientific exploration and regulatory measures, we can better safeguard human and environmental safety.
[1]https://www.inchem.org/documents/icsc/icsc/eics0561.htm
[2]https://cfpub.epa.gov/ncea/pprtv/documents/
[3]https://fscimage.fishersci.com/msds/86078.htm
[4]https://www.jaydinesh.com/guanidine-nitrate-uses/
[5 ] Hu Donglin. Research on wastewater treatment process based on guanidine nitrate [J]. Shanxi Chemical Industry, 2022, 42(02): 347-348+351. DOI: 10.16525/j.cnki.cn14-1109/tq.2022.02.132.
[6] Song Pingnan. Understanding guanidine nitrate and preventing its explosion [J]. Jilin Labor Protection, 2012, (03): 44.
[7] https://echa.europa.eu/registration-dossier/-/registered-dossier/13132/6/2/3
 |
 |
 |