-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

The decomposition behavior of guanidine nitrate has long been a significant research subject in the field of chemistry. By delving into its decomposition mechanisms, we can better understand its applications and impacts in fields such as explosives, chemical engineering, and safety production.
Due to its strong oxidizing nature, guanidine nitrate is classified as a Hazardous Material Class 5.1. It poses explosion risks when exposed to open flames, friction, impact, or heat. With a chemical formula of CH6N4O3, it contains nitro functional groups, rendering it highly unstable and prone to spontaneous decomposition even without external oxygen. This adds potential hazards during production, storage, and usage.
For instance, the major fire explosion accident at Hebei Kerui Chemical Co., Ltd. on February 28, 2012, resulted from the rapid decomposition and subsequent explosion of guanidine nitrate and incompletely reacted ammonium nitrate upon heating. This incident led to 25 deaths, 4 missing persons, and 46 injuries, releasing energy equivalent to 6.05 tons of trinitrotoluene (TNT). Hence, in-depth study of guanidine nitrate's thermal decomposition behavior holds crucial practical significance for safety production.
Guanidine nitrate appears as white granular solid, soluble in water and ethanol, sparingly soluble in acetone, with a melting point of 214~216°C. Its solubility in water increases with temperature: 12.5 g/100 g water at 20°C and 99 g/100 g water at 80°C. In methanol, solubility is 5.5 g/100 g at 20°C and 15.6 g/100 g at 60°C. Quality standard content is ≥98%. At what temperature does guanidine nitrate decompose? Hazard characteristics include strong oxidizing properties and explosion risks upon exposure to flames, friction, vibration, or impact, as well as decomposition at high temperatures, such as exploding when heated to 150°C.
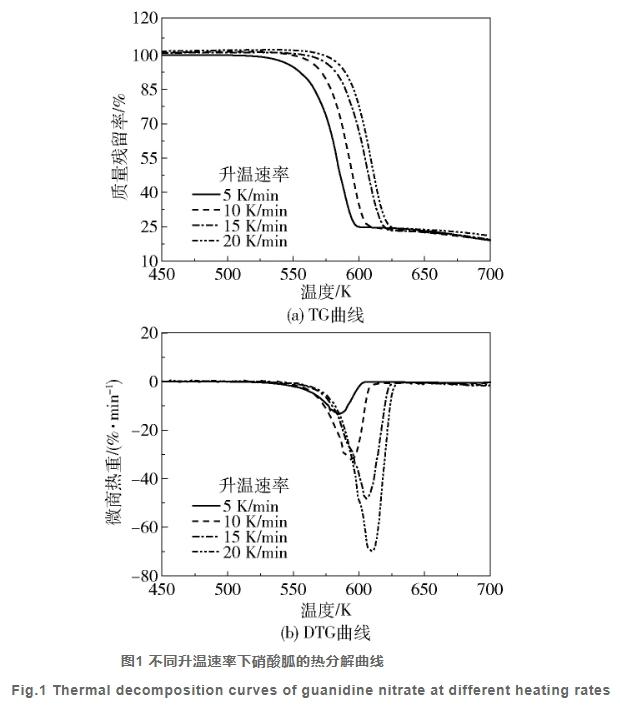
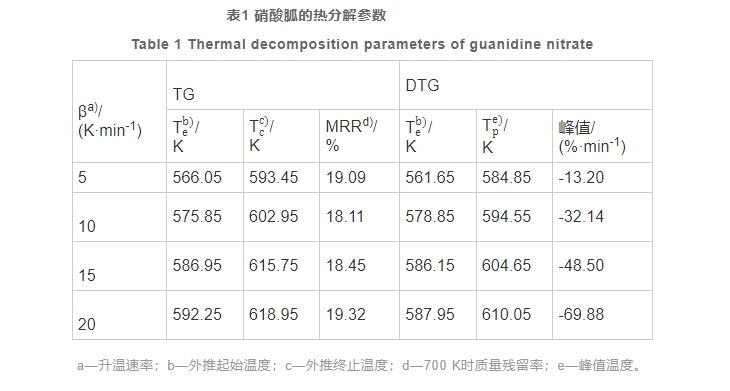
Figure 1 illustrates the thermogravimetry (TG) and differential thermogravimetry (DTG) curves of guanidine nitrate under heating rates of 5, 10, 15, and 20 K/min, while Table 1 presents the thermal decomposition parameters. It's observed that guanidine nitrate undergoes a single-step weight loss in the temperature range of 450~700 K, with an extrapolated onset temperature of 580.28 K and extrapolated end temperature of 607.78 K, representing its primary decomposition stage. Higher heating rates induce a lag effect, shifting the onset, peak, and end temperatures towards higher values, along with an increase in maximum decomposition rate.
Guanidine nitrate's decomposition process, characterized by explosiveness and toxic smoke emission, poses potential dangers. To ensure safety, it's crucial to identify potential hazards and take preventive measures. Firstly, understanding the decomposition products—nitric acid and nitrogen oxides—as irritants that can cause severe respiratory issues. Secondly, avoid friction, impact, or heat sources near guanidine nitrate, as these could trigger explosions.
Implement measures such as using designated explosion-proof containers and controlled temperature environments for storage and handling. Throughout the decomposition process, adherence to safety protocols is crucial. Personnel should wear appropriate personal protective equipment (PPE), including respirators, chemical-resistant gloves, goggles, and protective clothing. Additionally, establish clear leak response procedures and employ suitable neutralizers for immediate control and cleanup. Only trained professionals familiar with guanidine nitrate's Safety Data Sheet (SDS) should handle the material.
Compliance with regulations and adherence to industry standards are vital for safe decomposition. Familiarize with local, state, and federal regulations governing the storage, handling, and disposal of guanidine nitrate. Furthermore, adhere to industry standards established by organizations like the National Fire Protection Association (NFPA), which provide guidance for the safe handling of hazardous materials. By following these guidelines and implementing appropriate safety protocols, the risks associated with guanidine nitrate decomposition can be significantly reduced.
Guanidine nitrate's rapid decomposition at high temperatures releases a large volume of gas, making it valuable in various industrial applications:
(1) Pyrotechnics: Rapid gas release generates intense explosions, making it suitable for fireworks and special effects.
(2) Propellants: Under controlled conditions, guanidine nitrate decomposition can propel rockets and model aircraft due to high gas output.
The environmental impact of guanidine nitrate decomposition depends on its usage environment:
(1) Air Pollution: Guanidine nitrate explodes under friction, heat, or impact, igniting organic materials upon contact and releasing toxic vapors, including nitric acid and nitrogen oxides, upon decomposition. Improper combustion may also generate nitrogen oxides (NOx), leading to smoke and acid rain.
(2) Soil and Water Pollution: Accidental leaks or improper disposal can contaminate soil and water with nitrate, causing nitrate leaching that damages aquatic ecosystems and poses health risks to humans if it enters drinking water sources.
There's a growing focus on minimizing industrial processes' environmental impact:
(1) Closed Combustion Systems: In pyrotechnic technology, using closed combustion systems can capture and control nitrogen oxides, reducing air pollution.
(2) Improved Combustion Techniques: Optimizing propellant combustion processes ensures complete decomposition and minimizes nitrogen oxide formation.
(3) Alternative Propellants: Research into environmentally friendly propellants with similar performance characteristics is ongoing, including ammonium dinitramide (ADN) and nitrous oxide (N2O).
Through in-depth exploration of guanidine nitrate decomposition, we've enhanced our understanding of its chemical properties and behaviors. This not only helps us better utilize its characteristics but also guides us in more effectively managing and controlling its applications in engineering and safety fields. With ongoing research, we believe that understanding of guanidine nitrate decomposition will continue to improve, providing a more solid foundation for further developments in related fields.
[1] Yang Ran, Chen Dongliang, Zhang Dongsheng, et al. Thermal decomposition characteristics and kinetic analysis of guanidine nitrate [J]. Journal of Beijing University of Chemical Technology (Natural Science Edition), 2021, 48 (03): 1-8. DOI:10.13543/j.bhxbzr.2021.03.001.
[2] Wang Xuezhi. Study on the thermal runaway mechanism of guanidine nitrate [D]. China University of Petroleum (East China), 2017.
[3] Song Pingnan. Understanding guanidine nitrate and preventing its explosion [J]. Jilin Labor Protection, 2012, (03): 44.
[4] https://www.jaydinesh.com/guanidine-nitrate-uses/
[5] https://fscimage.fishersci.com/msds/86078.htm
 |
 |
 |