Methyldopa (English: Methyldopa), also known as α-methyl dopa, is a medication for treating hypertension. Its chemical name is L-2-amino-3-(3,4-dihydroxyphenyl)-2-methylpropanoic acid. The chemical structure is shown below:
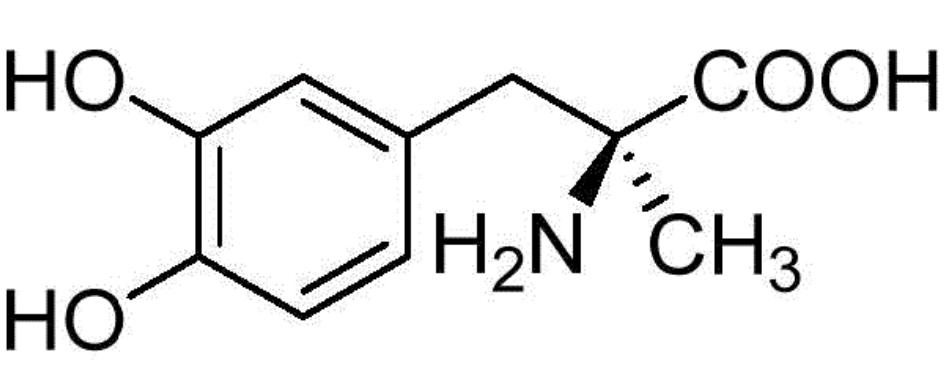
Methyldopa was discovered in the 1960s and is a classic drug for the treatment of hypertension. It is mainly used for moderate, mild or malignant hypertension, especially for renal hypertension and pregnancy-induced hypertension. It also has the effects of sedation and reducing intraocular pressure. It can be administered orally or intravenously. The drug takes effect in about 5 hours and lasts for about 1 day.
Methyldopa belongs to a general category of antihypertensive medications used to treat high blood pressure (hypertension). Hypertension adds stress to the heart and arteries, potentially leading to heart and artery dysfunction. This condition can damage blood vessels in the brain, heart, and kidneys, increasing the risk of stroke, heart failure, or kidney failure. Additionally, hypertension may increase the likelihood of a heart attack. However, effective blood pressure control can significantly reduce the risk of these health issues.
The antihypertensive effect of Methyldopa is mainly mediated by its pharmacologically active metabolite α-methyl norepinephrine, which acts as a central α-adrenergic receptor agonist. Stimulation of α-adrenergic receptors reduces peripheral sympathetic nerve activity and arterial pressure. Methyldopa causes a net reduction in the tissue concentrations of serotonin, dopamine, norepinephrine, and epinephrine. Overall, Methyldopa can reduce standing blood pressure, especially supine blood pressure, and occasionally causes symptomatic orthostatic hypotension. It also lowers plasma renin activity but has minimal impact on glomerular filtration rate, renal blood flow, or filtration fraction. There is no direct effect on cardiac function, although bradycardia may occur in some patients.
Methyldopa acts by controlling impulses along certain neural pathways, thereby relaxing blood vessels and making it easier for blood to pass through them. This helps to lower blood pressure. Methyldopa can only be used under a doctor's prescription.
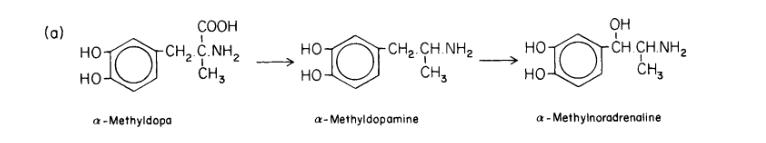
The exact methyldopa mechanism of action is not fully elucidated; however, the primary Methyldopa mechanism of action involves its effects on α-adrenergic receptors and aromatic L-amino acid decarboxylase, though to a lesser extent. Sympathetic output is regulated by α-2 adrenergic receptors and imidazoline receptors expressed on the adrenergic neurons of the medullary ventrolateral medulla. Methyldopa is metabolized by dopamine β-hydroxylase to α-methyl norepinephrine and then by phenylethanolamine N-methyltransferase to α-methyl epinephrine. The α-methyl norepinephrine and α-methyl epinephrine active metabolites mediate Methyldopa's therapeutic effects as agonists of presynaptic α-2 adrenergic receptors in the brainstem. Stimulation of α-2 adrenergic receptors inhibits adrenergic neuron output and reduces norepinephrine release in the brainstem. Thus, adrenergic signals to the peripheral sympathetic nervous system decrease, leading to lowered blood pressure.
The L-isomer of α-methyl dopa also lowers blood pressure by inhibiting aromatic L-amino acid decarboxylase (also known as DOPA decarboxylase), an enzyme responsible for the synthesis of dopamine and serotonin. Inhibition of this enzyme leads to the depletion of biogenic amines such as norepinephrine. However, the inhibition of aromatic L-amino acid decarboxylase has minimal impact on Methyldopa's antihypertensive effect.
After oral administration of Methyldopa, approximately 50% of the drug reaches peak plasma concentrations within 3 to 6 hours. Blood pressure typically reaches its maximum reduction effect within 4 to 6 hours. After intravenous administration, the hypotensive effect can last approximately 10 to 16 hours.
Methyldopa is lipid-soluble, able to cross the blood-brain barrier, and binds weakly to plasma proteins. In contrast, its main metabolites bind more strongly to plasma proteins. Acidic conjugates formed after oral administration are unstable, with only a small amount retained after intravenous administration. Its apparent volume of distribution ranges from 0.19 to 0.32 L/kg, with a total distribution volume of 0.41 to 0.72 L/kg.
Methyldopa is metabolized into the active metabolite α-methyl norepinephrine in the body. Additionally, the drug is extensively metabolized in the liver to its sulfate conjugates.
Approximately 70% of Methyldopa is excreted in the urine, including both the parent drug and metabolites. Unabsorbed drug is excreted via feces. Excretion is slower in patients with renal impairment, which may lead to accumulation of the drug and its metabolites in the body.
[1]Zhejiang Yefeng Pharmaceutical Co., Ltd. Methyldopa anhydrous crystalline form, solvate and preparation method thereof. 2023-03-07.
[2]https://zh.wikipedia.org/zh-cn/
[3]https://go.drugbank.com/drugs/DB00968
[4]https://www.mayoclinic.org/
[5]Gupta M, Al Khalili Y. Methyldopa[J]. 2019.
 |
 |
 |