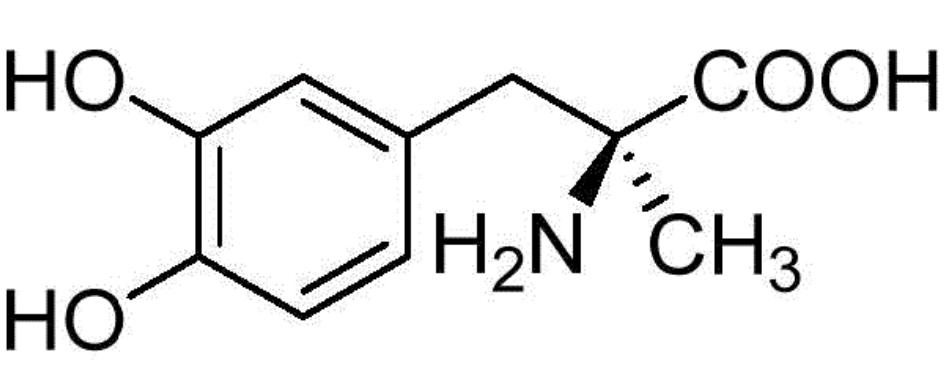
Methyldopa, or α-methyl-dopa, is a centrally acting sympatholytic and antihypertensive drug. It is an analogue of DOPA (3,4-dihydroxyphenylalanine) and is a prodrug, meaning it needs to be biotransformed into an active metabolite to exert its therapeutic effects. Methyldopa acts as an agonist on α(α)-2 adrenergic receptors, thereby inhibiting the outflow of adrenergic neurons and reducing vasoconstrictor adrenergic signals. There are two isomers of methyldopa: D-α-methyl-dopa and L-α-methyl-dopa, with the latter being the active form.
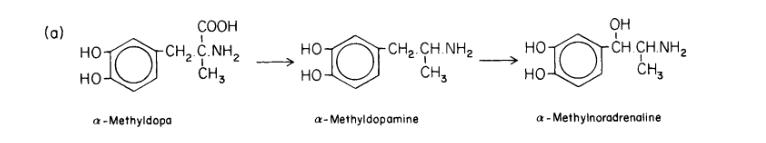
Methyldopa is an α-methyl analogue of dopamine, converted into α-methyl dopamine by dopamine decarboxylase, and then into α-methyl norepinephrine by dopamine-β-hydroxylase (as shown in the figure below). It is stored in vesicles and its release mechanism is similar to that of norepinephrine. However, α-methyl norepinephrine's potency at postsynaptic receptors is slightly lower than norepinephrine, suggesting that the false neurotransmitter action might be less significant in methyldopa's antihypertensive effects.
When first introduced, methyldopa was a primary medication for treating hypertension, but its use declined due to relatively severe side effects. Other safer and better-tolerated medications, such as α-receptor blockers, β-receptor blockers, and calcium channel blockers, became more common. Additionally, it has not been associated with reduced adverse cardiovascular events such as myocardial infarction and stroke, or with decreased overall mortality in clinical trials. Despite this, methyldopa remains a treatment option for pregnancy-induced hypertension (PIH) due to its relative safety during pregnancy compared to other antihypertensive drugs that may affect the fetus.
What is the drug methyldopa used for?
What is methyldopa used to treat? Methyldopa is widely used to treat hypertension. It is the preferred drug for managing pregnancy-induced hypertension and kidney impairment. Its mechanism of action involves conversion to α-methyl norepinephrine in central adrenergic neurons, which then stimulates central adrenergic receptors, particularly α-2 adrenergic receptors. Methyldopa uses is used clinically for:
Methyldopa does not cause birth defects and has been shown not to adversely affect the fetus. It maintains uterine perfusion and does not obstruct maternal cardiac output or renal or uterine blood flow. As an α-methylated derivative of dopamine (a natural precursor to dopamine and norepinephrine), Methyldopa uses include being a suitable alternative to clonidine for patients who cannot tolerate clonidine due to rebound hypertension or adverse effects. Although methyldopa was popular in the 1970s, other alternatives have replaced it as a primary antihypertensive medication. Methyldopa remains a second-line treatment option safe for use during pregnancy.
Methyldopa uses include its application in hypertensive emergencies. It is administered intravenously (as a parent drug ester), with a typical intravenous dose range (for α-methyl-dopa) of 20 to 40 mg/kg per day, given every 6 hours.
Methyldopa's antihypertensive effect is characterized by a relatively slow onset, with blood pressure reduction beginning approximately 2 to 3 hours after ingestion (whereas clonidine acts in 0.5 to 1.0 hours). Oral methyldopa reaches its lowest blood pressure point about 5 hours after administration, with effects lasting up to 24 hours. The usual starting dose of methyldopa is 250 mg twice daily, with a maximum dose of 3.0 grams. For patients with renal impairment, the dose should be reduced by half.
Methyldopa also effectively lowers supine blood pressure without causing orthostatic hypotension.
Methyldopa side effects
Rash
Rebound hypertension
Known hypersensitivity to any form of methyldopa
Methyldopa is commonly available in 125, 250, or 500 mg tablet forms. Fixed-dose combinations with thiazide diuretics are also available. The recommended dose for adults is 500 mg to 2 grams per day. If the patient experiences any adverse effects, the medication should be discontinued. Methyldopa can be administered intravenously as hydrochloride. Dilute the medication in 5% dextrose and add the required dose to 100 mL of 5% dextrose injection solution, and administer slowly over 30 to 60 minutes. Intramuscular or subcutaneous injection is not recommended due to unpredictable absorption.
[1]https://en.wikipedia.org/wiki/Methyldopa
[2]https://go.drugbank.com/drugs/DB00968
[3]https://www.sciencedirect.com/topics/neuroscience/methyldopa
[4]https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/methyldopa
[5]https://www.sciencedirect.com/science/article/abs/pii/B9780723612711500134
[6]https://www.ncbi.nlm.nih.gov/books/NBK551671/
[7]Gupta M, Al Khalili Y. Methyldopa[J]. 2019.
[8]https://www.drugs.com/
 |
 |
 |