Molybdic acid, also known by its English name molybdic acid, Molybdic acid cas no number is 7782-91-4 and molybdic acid formula is H2MoO4. Molybdenum (Mo, atomic number 42) is a biologically active transition metal in the fifth row (4d) of the periodic table. It was discovered by Carl Wilhelm Scheele in the form of molybdic acid (MoO3·H2O) and was first isolated as a dark metal powder by Peter Jacob Hjelm in 1781, although it does not exist as a free metal in nature.
Molybdic acid refers to the hydrated forms of molybdenum trioxide and related substances. The monohydrate (MoO3·H2O) and dihydrate (MoO3·2H2O) are well characterized. These are yellow paramagnetic solids.
Molybdic acid is the hydrated form of molybdenum trioxide and related substances, also known as molybdic hydroxide, molybdenum (VI) acid, or dihydroxyoxomolybdenum. Depending on the presence of water molecules in the compound, molybdic acid primarily exists as either a monohydrate or a dihydrate. The chemical formula for the monohydrate is MoO3·H2O, while the formula for the dihydrate is MoO3·2H2O. The chemical formula of molybdic acid is typically represented as MoO3·2H2O, where the molybdenum element exists in the form of the molybdate ion (MoO42-) and is combined with hydrogen ions (H+) and hydroxide ions (OH-).
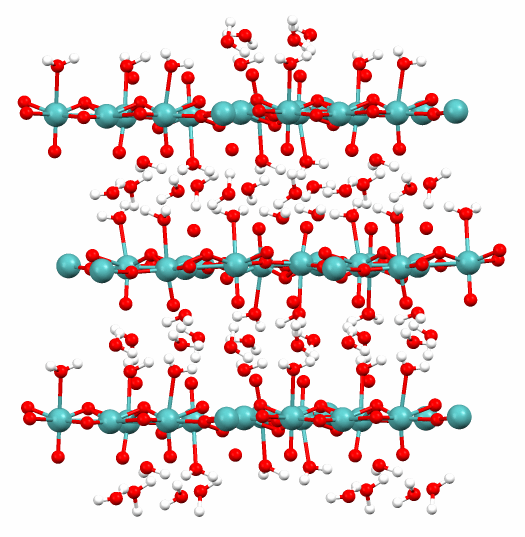
The solid form of molybdic acid is a coordination polymer. Its monohydrate MoO3·H2O consists of layers of octahedrally coordinated MoO5·(H2O) units, where four corners are shared. The dihydrate (as shown in the figure) has the same layered structure but includes "extra" H2O molecules between the layers.
In acidified molybdic acid aqueous solutions, the complex MoO3(H2O)3 can be observed. In this complex, the molybdenum atom adopts an octahedral molecular geometry, possibly bonding with three oxygen ligands and three water ligands. The salts of molybdic acid are called molybdates, which are formed by adding a base to the molybdic acid solution.
Catechol reacts with molybdic acid to form MoO2 ions and 1,2-cyclohexanedione. Molybdic acid is used as a heterogeneous catalyst and in the preparation of Froehde's reagent.
Molybdic acid precipitate is produced by reacting ammonium molybdate solution with nitric acid. The precipitate is then processed through washing, centrifugation, drying, and grinding.
Many molybdenum oxides are used as heterogeneous catalysts, such as in oxidation reactions. Molybdic acid and its salts are used in the preparation of Froehde's reagent for the presumptive identification of alkaloids.
Molybdic acid plays a crucial role in both industrial and laboratory settings, with wide applications in analytical chemistry and catalytic reactions. Its unique chemical properties make it an ideal choice for many important reactions. Given these advantages, readers may consider incorporating molybdic acid into their relevant industrial processes or laboratory work to meet specific needs.
 |
 |