-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

Oleyl alcohol (Oleyl alcohol synonyms cis-9-octadecen-1-ol) is an unsaturated fatty alcohol with the chemical formula C18H36O. Its structure is CH3(CH2)7–CH=CH–(CH2)8OH, and it has a CAS number of 143-28-2. Oleyl alcohol is a versatile unsaturated fatty alcohol used in various industries, including plastics and surfactants, as well as in metalworking, petrochemicals, pulp and paper, paints and coatings, where it serves as a thickener, emulsion stabilizer, coupling agent, petroleum additive, pigment dispersant, rust inhibitor, wetting agent, defoamer, metal cutting lubricant, plasticizer, fabric softener, detergent, mold release agent, co-solvent, softener, tackifier, and spreading agent.
Oleyl alcohol features an 18-carbon straight-chain hydrocarbon with a hydroxyl group (OH) at one end and a cis double bond (C=C) between the 9th and 10th carbon atoms. This unique structure imparts significant hydrophobicity to oleyl alcohol, making it insoluble in water but interacting well with lipids and oils. The presence of the double bond causes the oleyl alcohol molecule to adopt a bent shape, contrasting with the linear structure of saturated alcohols, thereby affecting its molecular arrangement and properties. Additionally, the combination of the hydrophobic hydrocarbon chain and the hydrophilic hydroxyl group gives oleyl alcohol a certain amphiphilic characteristic, allowing it to interact with both water and oil. This feature is particularly important for its use as an emulsifier.

Oleyl alcohol structure
The HLB (Hydrophilic-Lipophilic Balance) value is a measure used to characterize the hydrophilicity or lipophilicity of a substance. It reflects the balance between the hydrophilic and lipophilic groups in a surfactant, calculated by the formula HLB = Hydrophilic molecule weight / Lipophilic molecule weight. Surfactants are molecules with both hydrophilic and lipophilic parts, and their HLB value indicates the relative size and strength of these parts. A higher HLB value suggests greater hydrophilicity, while a lower value indicates stronger lipophilicity. Typically, HLB values range from 1 to 40. In practical applications, an HLB value less than 10 indicates stronger lipophilicity, while a value greater than 10 indicates stronger hydrophilicity. The balance point for hydrophilic and lipophilic properties is usually set at an HLB value of 10.

In its most basic form, the HLB system is calculated using the following formula:
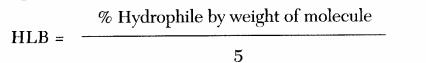
Oleyl alcohol is a fatty alcohol with a long hydrocarbon chain. Its HLB (Hydrophilic-Lipophilic Balance) value is not always easy to find because it is not a standard surfactant but a fatty alcohol used in various formulations due to its skin-conditioning and emulsifying properties. Studies have reported that oleyl alcohol (5eo), with a total molecular weight of 489 and a hydrophilic molecular weight of (5)(44)=220, has a hydrophilicity of 45.0%. Dividing by 5, the HLB of oleyl alcohol equals 9.0.
Oleyl alcohol is prepared by mixing oleic acid ethyl ester with anhydrous acetic acid and then rapidly adding metal sodium pieces. The reaction starts vigorously and gradually stabilizes. Next, anhydrous ethanol is added and heated until the metal sodium completely reacts. Water is then added for refluxing for 1 hour to ensure that any unreacted oleic acid ethyl ester undergoes saponification. After the reaction is complete, the mixture is cooled, extracted with ether, and then neutralized, washed, and dried. After evaporating the ether, a reduced pressure distillation is carried out to collect fractions within the range of 150-152°C (0.133 kPa), yielding oleyl alcohol. The process has a yield of approximately 50%.
As an efficient fatty alcohol with excellent lubricating and surfactant properties, oleyl alcohol is widely used in cosmetics, pharmaceuticals, and other industrial fields, demonstrating significant advantages in enhancing product performance. If you are interested in the potential applications of oleyl alcohol, it is recommended to visit Guidechem to find professional suppliers and obtain more product information and purchasing options. Through Guidechem, you can easily locate reliable oleyl alcohol suppliers to meet your specific needs and further explore this valuable chemical.
[1] Chang Ying, Wang Chenghe, Liu Yanjun. Synthesis and application of oleyl alcohol[C]. //2003 Surfactant Technology and Economics Collection. Dalian: Fine and Specialty Chemicals Editorial Department, 2003:199-203.
[2] Liu Yanjun, Chang Ying. Synthesis and application of oleyl alcohol[J]. Fine and Specialty Chemicals, 2004, 12(5): 8-11. DOI: 10.3969/j.issn.1008-1100.2004.05.002.
[3]https://en.wikipedia.org/wiki/Oleyl_alcohol
[4]https://baike.baidu.com/item/%E6%B2%B9%E9%86%87
[5]https://baike.baidu.com/item/HLB
[6]O'Lenick Jr A J, Parkinson J K. Three-dimensional HLB[J]. Cosmetics and toiletries, 1996, 111(10): 37-46.
 |
 |
 |