-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

Synthesizing Vindoline is a challenging and significant area of chemical research, garnering continuous attention for its methods and techniques. Through ongoing experiments and exploration, scientists have gradually revealed the synthetic pathways of Vindoline, providing crucial references and guidance for its industrial production and drug development.
Vincamine and Vincristine are widely recognized members of the indole alkaloid family, primarily used as anticancer drugs. To date, they are mainly extracted from Catharanthus roseus, as this plant is their sole natural source. However, due to their low concentrations in the plant (in the range of parts per hundred thousand to million), chemical synthesis is economically unfeasible, leading to high market prices. Despite their dose-dependent neurotoxic side effects, limiting their applications, efforts by medicinal chemists to synthesize them chemically or modify their structures for more efficient and less toxic analogs persist. Biologists have also explored plant tissue culture methods for their production.
Since the 1960s, research on indole alkaloid production via Catharanthus roseus tissue culture has been ongoing. While studies in the late 1970s focused on using this method to produce these alkaloids, until now, only ajmalicine, serpentine, and catharanthine can be produced, and industrial production remains unrealized. As Vindoline is an important precursor in the semi-synthesis of Vincamine or Vincristine, its large-scale production through plant cell culture can enable semi-synthesis. Thus, once two raw materials, catharanthine and Vindoline, are produced in large quantities through plant cell culture, Vincamine can be semi-synthesized. The structure of Vindoline is depicted in the figure below.
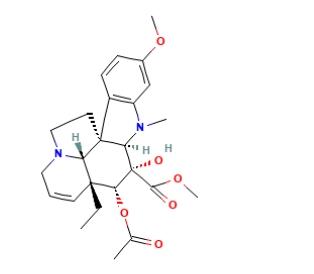
Vindoline can be synthesized through two main methods:
(1) Cyclization Reactions: These reactions form new rings by connecting two molecules. The Boger synthesis is a notable example, employing a tandem [4+2]/[3+2] cyclization cascade to simultaneously generate three rings in the core structure of Vindoline.
(2) Stereoselective Reactions: Vindoline has multiple stereocenters (atoms with specific spatial arrangements). Chemists utilize various techniques during synthesis to ensure the correct three-dimensional arrangement of these centers. Strategies such as enantioselective catalysis and chiral starting materials are employed for this purpose.
(3) Functional Group Manipulation: Throughout the synthesis process, chemists introduce various functional groups (e.g., carbonyl, ether) and strategically manipulate them through reduction, elimination, protection/deprotection reactions to obtain the final structure of Vindoline.
(1) Metabolic Engineering: Scientists introduce genes from the Vindoline biosynthetic pathway of Catharanthus roseus (Madagascar periwinkle) into yeast. This endows yeast with the enzymatic ability to convert simpler precursors like tabersonine into Vindoline.
Compared to complex chemical synthesis, biosynthesis offers a potentially more sustainable and scalable method for Vindoline production.
Vindoline biosynthesis is a complex natural process discovered in Catharanthus roseus. It involves multiple enzyme-catalyzed steps starting from the terpenoid intermediate strictosidine. Key enzymes include:
- Geraniol Synthase (GES)
- Geraniol 8-Hydroxylase
- 8-Hydroxygeraniol Oxidoreductase (GOR)
- Secologanin Synthase
- Tabersonine 3-Hydroxylase
- Tabersonine 3-Reductase
- 3-Hydroxy-16-methoxy-2,3-dihydrotabersonine-N-methyltransferase (NMT)
- Desacetoxyvindoline-4-Hydroxylase (D4H)
- Deacetylvindoline-4-O-Acetyltransferase (DAT)
These enzymes work together to convert secologanin to Vindoline through a series of hydroxylation, oxidation, cyclization, methylation, and acetylation reactions.
In this context, biocatalysts refer to the enzymes mentioned above. Each enzyme, acting as a catalyst, is a specific protein that accelerates specific chemical reactions without being consumed. They play crucial roles in Vindoline synthesis:
(1) Selectivity: Enzymes are highly selective, meaning they only act on specific substrates and promote the desired reactions, minimizing unnecessary side reactions and allowing for efficient Vindoline formation. Compared to traditional chemical synthesis, enzymes work under mild conditions (temperature, pressure, pH), reducing energy consumption and making the process more environmentally friendly.
(2) Stereocontrol: Many enzymes can control the stereochemistry of product molecules. Vindoline possesses a specific 3D structure with multiple chiral centers. Enzymes ensure the correct arrangement of these centers, crucial for the biological activity of the final product.
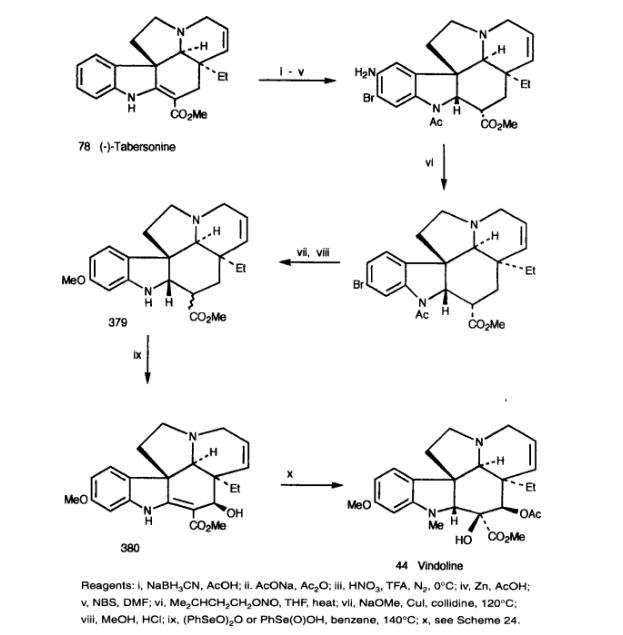
Danieli et al. developed a method to convert abundant tabersonine into 11-methoxytabersonine, then into Vindoline. Due to the ineffective and clean electrophilic substitutions of tabersonine and 2,16-dihydroxytabersonine, N-acetyl-2,16-dihydroxytabersonine was selected as the substrate. The nitration reaction yielded the 10-nitro derivative in high yield, which was then converted to 11-methoxy-2,16-dihydroxytabersonine (379) through a standard process. Attempting to dehydrogenate the product 380 with benzyl selenide unexpectedly introduced the desired C-17 β-hydroxy group. The product 380 was then transformed into Vindoline via the previously described Catharanthine synthesis method.
(1) Acquisition of Catharanthus roseus Callus
Catharanthus roseus young stems were used as explants, surface sterilized in 70% ethanol for 30-50 seconds, then subjected to secondary sterilization in 5-15% sodium hypochlorite solution for 10-20 minutes. After rinsing three times with sterile water, the stems were cut into 2-millimeter segments in a laminar flow hood. These segments were then inoculated into dedifferentiation culture medium and cultured at approximately 25°C under 2000 lx light intensity for 12-16 hours per day for 3-4 weeks to obtain white callus tissue. The callus tissue was further subcultured in the mentioned medium for more than three passages, selecting for fast-growing, soft-textured, and stable cell lines.
(2) Preparation of Dedifferentiation Culture Medium
Indole-3-acetic acid (IAA) at 1.0 mg/L and cytokinin 6-benzylaminopurine (6-BA) at 1.0 mg/L were added to Murashige and Skoog (MS) medium supplemented with 20 g/L sucrose. The pH was adjusted to 5.8 with acid or alkali, followed by addition of 6.5 g/L agar. The medium was autoclaved at 115°C under 0.1 MPa pressure for 15 minutes and then cooled to prepare slanted dedifferentiation culture medium.
(3) Subculture of Catharanthus roseus Callus
The induced callus cell lines were subcultured more than three times in the mentioned subculture medium. The subculture conditions were 20°C, avoidance of light, and a subculture period of 3 weeks. During subculture, loose-structured and fast-growing callus tissues were selected for further subculture.
(4) Induction of Catharanthus roseus Polyploid Cells
Catharanthus roseus callus tissue obtained from the previous steps was inoculated into induction culture medium containing 10 mg/L colchicine. The induced culture was shaken at 100 rpm at 20°C in the dark for 7 days. The cells were then collected and subcultured in colchicine-free subculture medium for at least three passages. The resulting fast-growing cell lines (with growth rates exceeding 5 times the initial inoculum per culture period) were identified as mixed polyploid cell lines via microscopic examination after aceto-orcein or silver staining.
(5) Expansion Culture of Catharanthus roseus Polyploid Cells
The vigorous-growing polyploid cell lines obtained from the previous step were inoculated onto expansion culture medium. After culturing at 25°C in the dark for 3 weeks, the cell cultures with more than 10 times the initial inoculum were obtained.
The vigorously growing polyploid Catharanthus roseus cell lines were inoculated into synthesis medium at a concentration of 200 g/L. The suspension cultures were shaken at 90 rpm at 25°C in the dark or in a large-scale bioreactor for 7 days to obtain cells rich in Vindoline. After harvesting the cells, the culture medium could be adjusted and reused in the bioreactor.
Synthesis Medium Preparation: Sucrose at 30 g/L, indole-3-acetic acid (IAA) at 1 mg/L, and kinetin (KT) at 1 mg/L were added to modified MS medium. The pH was adjusted to 5.8 with acid or alkali, followed by addition of 4.5 g/L agar. The medium was autoclaved at 115°C under 0.1 MPa pressure for 15 minutes and then cooled. After cooling, filtered and sterilized precursor compounds (acetyl coenzyme A at 10 μg/L, phenylalanine at 0.5 μmol/L, tryptamine at 5 μmol/L, acetic anhydride at 10 μl/L, and dithiothreitol at 10 mg/L) were added, and the pH was adjusted to 5.8 with sterile acid or alkali to prepare the synthesis medium.
The cells enriched with Vindoline synthesized during cultivation are subjected to extraction by placing them in an extraction tank, followed by high-speed agitation (300 rpm) to disrupt the cells. A 95% ethanol solution, with a volume of 1 ml/g of fresh cell weight, is added for extraction at room temperature. The crude extract is obtained by vacuum concentration (0.01 MP), followed by the addition of twice the volume of ethyl acetate. After adjusting the pH to 8.0 with sulfuric acid and rapid shaking, the alkaloids are extracted three times. The mixture containing Vindoline is obtained after vacuum drying. High-performance liquid chromatography (HPLC) is employed to determine the Vindoline content in this mixture, revealing a concentration of 1 mg/g of cell dry weight.
The primary application of synthesized Vindoline lies in its role as a precursor to important anticancer drugs, vincristine and vinblastine. The low natural abundance of these alkaloids in Catharanthus roseus plants, coupled with the difficulty in their chemical synthesis, makes synthesized Vindoline a valuable alternative for large-scale production of these life-saving drugs.
Beyond its established role as a precursor, research is underway to explore potential applications of synthesized Vindoline:
Antidiabetic Properties: Studies suggest that Vindoline may possess antidiabetic properties, although further research is needed to confirm its effectiveness as a therapeutic approach.
Drug Development: The unique chemical structure of synthesized Vindoline holds promise for developing novel drugs with diverse therapeutic applications. By modifying the Vindoline molecule, scientists may create drugs targeting various diseases.
In conclusion, the synthesis of Vindoline represents a challenging and forward-looking research field. Continual refinement and optimization of synthesis methods will provide crucial technical support and guidance for drug development and production. It is hoped that this article inspires interest and exploration in the field of Vindoline synthesis.
[1] https://pubchem.ncbi.nlm.nih.gov/compound/260535
[2] https://www.sciencedirect.com/topics/chemistry/vindoline
[3] https://www.nature.com/articles/s42003-021-02617-w
[4] https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2531198/
[5] Tsinghua University. A method for producing Vindoline. 2008-12-31.
 |
 |