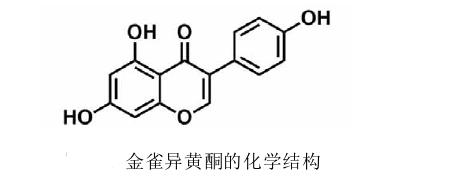
Genistein is a naturally occurring compound belonging to the isoflavone class of compounds, with a chemical structure of C15H10O5. It is described as an angiogenesis inhibitor and a phytoestrogen. This article provides valuable insights into the diversity of genistein and its impact on health, from understanding its origins to exploring its uses and potential side effects.
Genistein, an isoflavone, was initially isolated from the dyer's broom plant Dyer's Genista tinctoria L. It is widely distributed in leguminous plants. Isoflavones like genistein and daidzein are found in many plants, including clover, fava beans, soybeans, kudzu root, and the primary food source buckwheat, as well as in medicinal plants such as licorice and large-leaved kuding tea, and coffee. It can also be found in Amur cork tree cell cultures. As an isoflavone, mammalian genistein exhibits estrogen-like functions. Several biological effects of genistein have been reported in preclinical studies, such as antioxidant, anti-inflammatory, antimicrobial, antiviral activities, angiogenesis, estrogenic effects, and pharmacological activities related to diabetes and lipid metabolism.
The biological effects of genistein in the body can be divided into estrogen receptor-dependent effects and estrogen receptor-independent effects. Estrogen receptor-dependent effects occur through binding with the respective estrogen receptors α and β (ERα, ERβ). It is believed that the estrogenic effects of plant estrogens are bidirectional and concentration-dependent, exhibiting weak estrogenic activity at low concentrations (10-8 to 10-5 M) but antiestrogenic activity at high concentrations (10-5 to 10-4 M). The action of genistein at the cellular or molecular level is influenced by multiple factors, including but not limited to concentration dependence, receptor status, endogenous estrogen levels, and the type of target organ or cell. In intact ovarian function and normal endogenous estrogen levels, low concentrations of genistein can compete for binding sites with estrogen-binding globulin in the serum, increasing the concentration of free estradiol in the serum, enhancing estradiol effects, and thus exhibiting estrogenic activity, while high concentrations of genistein can not only bind to estrogen-binding globulin but also competitively bind to estrogen receptors to produce antiestrogenic effects. When endogenous estrogen levels are extremely low (such as after ovariectomy or menopause), genistein can bind more to estrogen receptors and produce estrogenic effects. However, the estrogenic effects of genistein vary among tissues, i.e., it may exhibit estrogenic effects or antiestrogenic effects on different tissues, or the intensity of estrogenic effects may vary among different tissues, making genistein a selective estrogen receptor modulator. Estrogen receptor-independent effects are biological effects of genistein produced through pathways other than ERα and ERβ, such as affecting neurotrophic factor-regulated cell proliferation, blocking protein tyrosine kinase, activating the MAPK signaling pathway, etc.
The activity of plant estrogens is influenced by their affinity with the respective receptors, but affinity does not fully correlate with the effects produced. The affinity of genistein with ERα is 25 times lower than that of 17β-estradiol, while its affinity with ERβ is comparable to that of 17β-estradiol (Kuiper et al. Gs's affinity with ERβ is about one-third of that of 17β-estradiol, i.e., 36:100). However, at the same concentration as 17β-estradiol (1000 nM), the gene activation effects of genistein through ERα and ERβ are approximately twice that of 17β-estradiol. Like other plant estrogens, genistein has a much higher affinity for ERβ than for ERα, about 22 times that of the latter. This is also one of the mechanisms of genistein as a selective estrogen receptor modulator.
Although the pharmacological mechanisms of genistein are not yet fully understood, it has been widely accepted as a phytoestrogen. It is mainly used in the following aspects: first, prevention and treatment of certain hormone-dependent tumors such as breast cancer, prostate cancer, and colorectal cancer. Second, alleviation of menopausal symptoms such as hot flashes, vaginal dryness, etc. Third, treatment of postmenopausal osteoporosis. Fourth, prevention and treatment of atherosclerosis and coronary heart disease. Fifth, improvement of learning and memory, prevention of senile dementia.
So far, no evidence has been found of significant short-term or long-term adverse effects of consuming plant estrogens on the human body. Animal studies have shown that sheep grazing on pastures rich in plant estrogens may develop infertility, a phenomenon that can be explained by the antiestrogenic effects of plant estrogens. Busby et al. conducted drug trials on 30 healthy adult men with a single oral dose of genistein at a dose of 16 mg/kg body weight, and the participants showed no significant behavioral or physiological abnormalities. Chemical structure analysis showed the presence of a 0.6 nm long biophore in the structures of 17βE2 and hexestrol, which has been confirmed to have carcinogenic effects. Cunningham et al. used a computer-aided evaluation system to analyze and found that genistein and other plant estrogens do not contain this structure, thus suggesting that genistein does not possess the carcinogenic properties of substances like estradiol.
Genistein possesses antioxidant properties, meaning it can help neutralize harmful free radicals in the body, reducing oxidative stress and inflammation.
As a plant estrogen, genistein can mimic the activity of endogenous estrogens in the body. This property may help control symptoms associated with hormonal imbalances, such as menopausal symptoms and osteoporosis.
Research suggests that genistein may promote cardiovascular health by improving vascular function, lowering cholesterol levels, and reducing blood pressure, potentially lowering the risk of heart disease.
The potential role of genistein in cancer prevention has been studied. Some research indicates that it may inhibit the growth of cancer cells, especially breast cancer, prostate cancer, and colon cancer, although more research is needed to confirm these effects.
Genistein may help support bone health by promoting bone formation and inhibiting bone resorption. This makes it potentially beneficial for preventing osteoporosis and maintaining overall bone density.
Genistein was first isolated from the dyer's broom (Genista tinctoria) in 1899. The structure of this compound was determined in 1926 when it was found to be identical to that of pr
unetol. It was chemically synthesized in 1928. It has been shown to be a major secondary metabolite of clover and glycine.
Genistein is a potent isoflavone found in various plant-based foods. When this compound occurs in glycoside form, it is sometimes referred to as genistin, commonly found in soybeans and other legumes. The exploration of alternative names for genistein highlights its diversity in nature and its importance in traditional medicine and scientific research. Throughout history, foods and herbs rich in genistein have played significant roles in various cultures, especially in Asian cuisine and traditional Chinese medicine. In addition to culinary uses, the medicinal value of genistein has been recognized for centuries, with historical records documenting its potential health benefits and therapeutic applications.
Genistein has been shown to be effective against various types of human cancers, such as breast cancer, lung cancer, liver cancer, prostate cancer, pancreatic cancer, skin cancer, cervical cancer, bone cancer, uterine cancer, colon cancer, kidney cancer, bladder cancer, neuroblastoma, stomach cancer, esophageal cancer, pituitary cancer, salivary gland cancer, testicular cancer, and ovarian cancer. The primary anticancer mechanisms of genistein are summarized as follows:
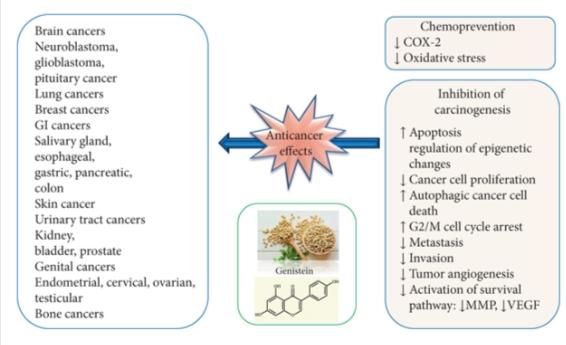
The main molecular targets and signaling pathways of genistein as a potential anticancer agent are shown below:
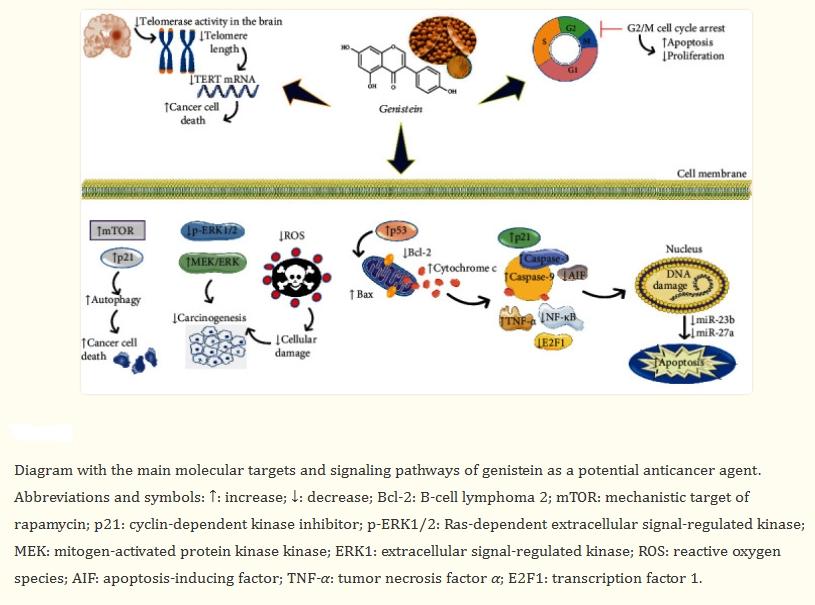
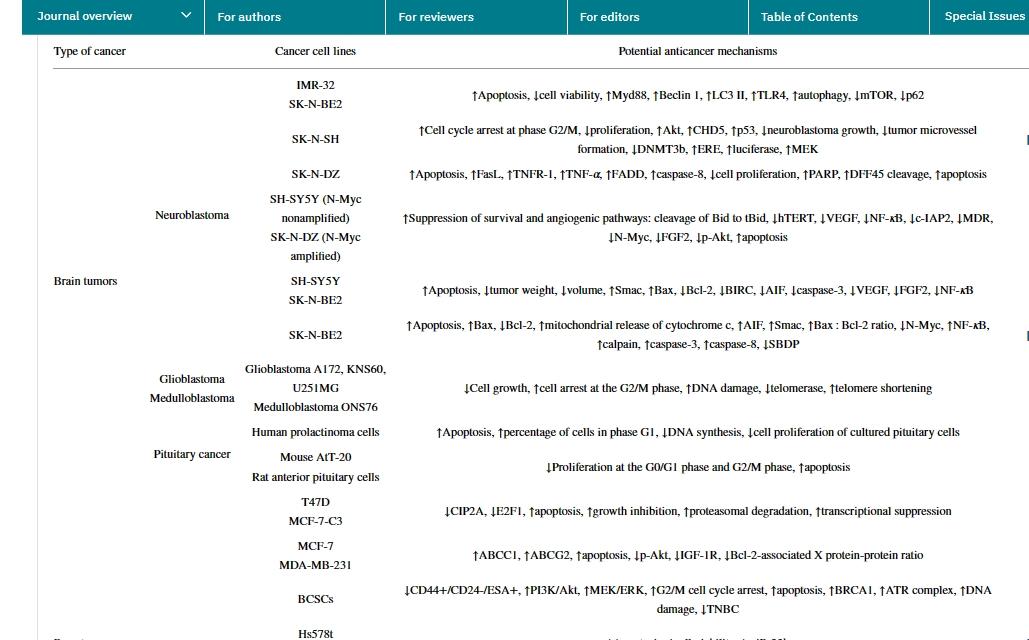
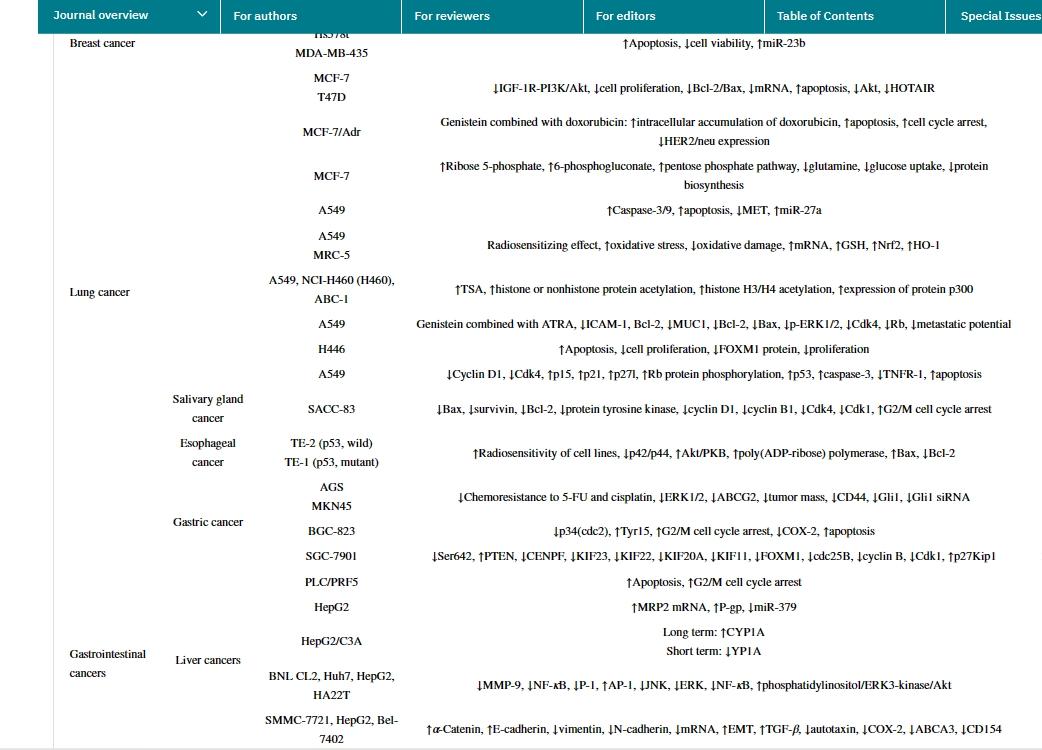
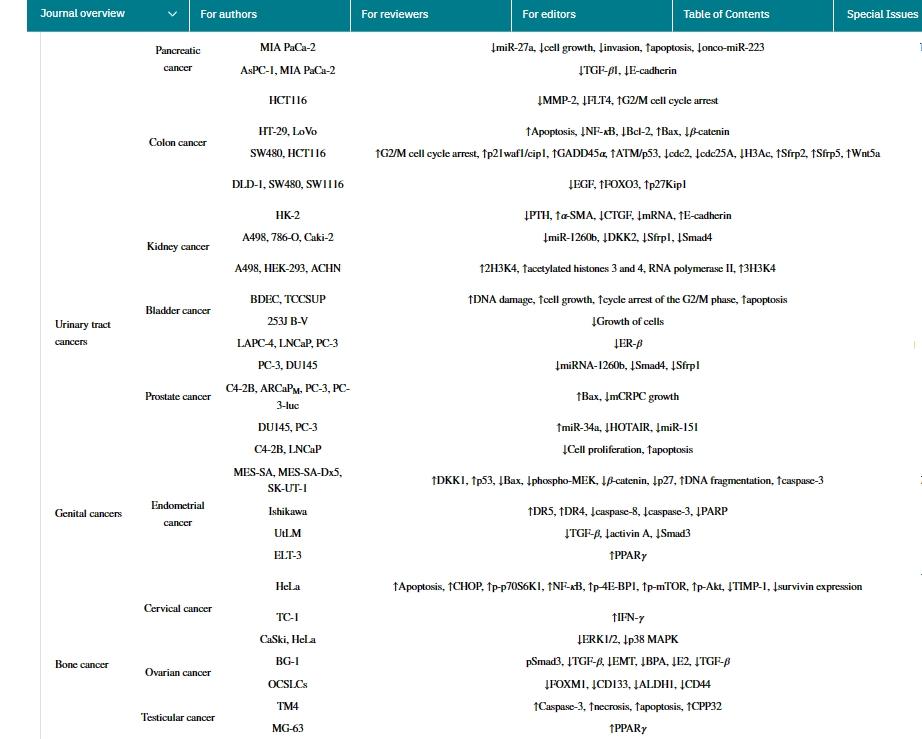

Genistein possesses various biological activities. As a plant estrogen, it acts as an agonist or antagonist of estrogen in mammals. However, the health properties of genistein seem to extend beyond their estrogenic activity. It has been reported that genistein has numerous preclinical pharmacological activities, such as antioxidant, anti-inflammatory, and antibacterial activities, angiogenesis and estrogenic effects, as well as pharmacological activities related to diabetes and lipid metabolism. However, the most researched activity is anticancer and cytotoxic activity. In human cancers, genistein stimulates CIP2A downregulation, significantly reduces the number of cells, induces cell arrest, and upregulates TNFR-1 protein and mRNA expression. It also activates p53 protein and caspase-3 and caspase-10. Genistein inhibits HSC activation, reduces α-SMA expression and collagen matrix accumulation, and increases serum ALT and AST levels. Additionally, genistein downregulates TGF-β expression and stimulates TGF-β/Smad signaling. In clinical studies of various cancer types, the anticancer effects of genistein have been confirmed, with the most significant anticancer effects observed in breast cancer and prostate cancer. In the future, more clinical trials and research using genistein formulations with higher bioavailability will open up new prospects in the research field.
[1] https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8315847/
[2] https://www.sciencedirect.com/topics/neuroscience/genistein
[3] https://en.wikipedia.org/wiki/Genistein
[4] Zhu JH, Xu J. Research Progress on the Anti-Aging Effect of Genistein in the Central Nervous System[J]. Journal of Sichuan Anatomy, 2003, (04): 34-37.
 |
 |
 |