What is the difference between dioscin and diosgenin? Dioscin and diosgenin are two common phytochemical constituents that bear strikingly similar names, which can easily lead to confusion. However, they possess certain differences in chemical structure and biological activity. Understanding the distinction between these two is crucial for a proper grasp of their functions and applications. In this article, we delve into the dissimilarities between dioscin and diosgenin, aiming to provide readers with a clearer comprehension.
Dioscin is a typical natural saponin, while diosgenin is a typical sapogenin. These two components are widely present in the Dioscoreaceae, Liliaceae, and Solanaceae families. Among these plants, Dioscoreaceae plants are renowned for their abundant dioscin and diosgenin, such as Japanese yam. In the 1960s, it was first discovered in the former Soviet Union that saponin components have anti-atherosclerotic effects. Increasing evidence suggests that dioscin and diosgenin can prevent myocardial ischemia and angina pectoris, and prevent myocardial infarction. The DiaoXinXinXin capsule recorded in the Chinese Pharmacopoeia is an extract of Japanese daylily and Pan Tai Gu Cao. It has the effects of promoting blood circulation, removing blood stasis, invigorating qi, relieving pain, dilating coronary arteries, and improving myocardial ischemia. It is used for the prevention and treatment of coronary heart disease (National Pharmacopoeia Commission, 2015), demonstrating the enormous potential of dioscin in cardiovascular disease protection.
Diosgenin is a major bioactive component in many edible legumes and roots, especially in fenugreek seeds and wild yam rhizomes. Diosgenin is found in 137 different species of the Dioscorea genus. Among them, 41 species contain more than 1% diosgenin. It is sourced from the roots of Dioscorea polystachya and is typically used as a precursor for producing synthetic steroid chemicals (such as progesterone and cortisol).
Glycosides are formed when a glycone (referred to as a glycone) is linked to one or more sugar groups through a glycosidic bond. Minor variations in compound structure are believed to significantly affect their biological activity. Therefore, different substituents, sugar chain compositions, and chemical bonds can influence the biological activity of dioscin and diosgenin.
There are many structural analogs of diosgenin. Diosgenin is obtained by linking the trisaccharide α-L-Rha-(1->4)-[α-L-Rha-(1->2)]-β-D-Glc to position 3 of diosgenin through a glycosidic bond; 13 can be hydrolyzed to convert into diosgenin. Considering the chemical structure of diosgenin and its impact on lipid bilayer membranes similar to cholesterol, they play a crucial role in cholesterol metabolism, namely inhibiting the absorption of dietary cholesterol by the intestines and accelerating the conversion of cholesterol into bile acids. Methyl original potato saponin (MPD), pseudo-original potato saponin (PPD), original potato saponin (PD), shanyun saponin, and tomato alkaloid are other analogs of diosgenin, which have similar pharmacological effects and are mainly extracted from the rhizomes of Dioscorea, Trigonella, Dioscorea, Dioscorea, and Dioscorea, among which Dioscorea contains the highest content. The molecular formula, molecular weight, source, and chemical structure of diosgenin and its main analogs are shown in the table.
Diosgenin (25R-spirost-en-3β-ol) is a C27-type triterpenoid spirostane glycoside, with a molecular weight of 414.62 and a molecular formula of C27H42O3 (see figure below). Structurally, it is a spirostane steroid with a hydroxyl group at the β position. It also has a double bond at positions 5 and 6 and an R configuration at position 25. It has a hydroxyl group at position three; the hydroxyl group is typically bound to sugar, imparting water solubility and high saponification to the compound. Its solubility in water is approximately 0.7°ng/ml. It is a white crystalline powder, soluble in ethanol, DMSA, and dimethylformamide, and other organic solvents. Cholesterol is the precursor of diosgenin biosynthesis, catalyzed by two P450 enzymes: c-16,22-hydroxylase and C-26 hydroxylase.

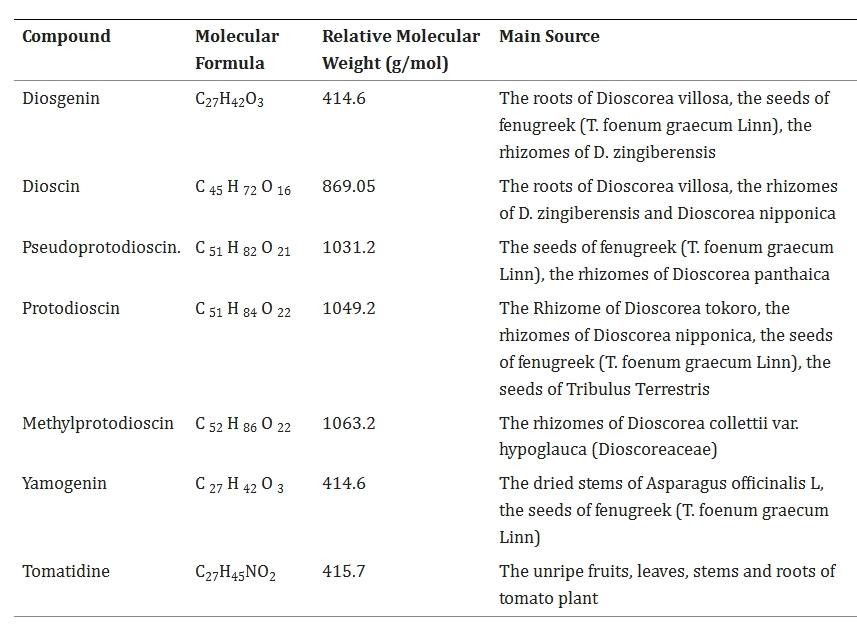
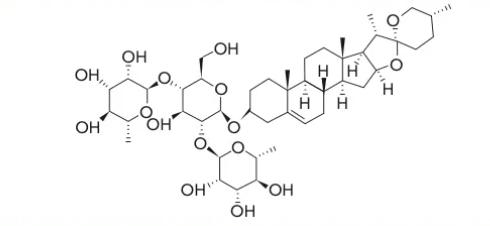
Dioscin (C45H72O16) is a natural steroid saponin composed of a spiropentane glycoside diosgenin, and its sugar chain is connected to the 3-hydroxy group through a glycosidic bond. Its chemical name is (3β,25R)-spirost-5-en-3-ylO-6-deoxy-α-L-mannopyranosyl-(1–2)-[6-deoxy-α-L-mannopyranosyl-(1–4)]-β-D-glucopyranoside. The structure is as follows:
The structures of the two are similar. Dioscin is a glycoside, meaning it is a molecule where a sugar unit (glucose) is linked via a glycosidic bond to another molecule (diosgenin). Diosgenin, on the other hand, is a sapogenin, referring to the aglycone component obtained from hydrolyzing (removing) the sugar unit from dioscin. Diosgenin possesses a steroidal sapogenin structure.
Studies suggest that dioscin may have lower bioavailability compared to diosgenin. This implies that the efficiency of dioscin absorption by the human body might be reduced.
Both dioscin and diosgenin exhibit a range of biological activities, especially concerning heart health. However, due to structural differences, mechanisms may slightly vary.
Dioscin serves as a precursor to steroid hormones, such as cortisone, and can be used to treat various ailments, including arthritis, asthma, and eczema. Research is also underway to explore its potential in treating cancer, Alzheimer's disease, and other conditions.
Dioscin is sometimes added as a natural health supplement to foods and dietary supplements. However, limited scientific evidence supports the use of dioscin for most of these purposes.
Dioscin is occasionally used in cosmetics as an anti-aging agent or skin whitening agent. However, further research is needed to confirm these effects.
In the pharmaceutical industry, dioscin serves not only as a vital starting material for several steroid drugs but also exhibits significant potential and interest in treating various diseases such as cancer, diabetes, arthritis, asthma, and cardiovascular diseases. Supplementing dioscin is considered an excellent way to promote women's health as it can slow the decline in estrogen and progesterone levels, helping to prevent hormonal imbalances. Essentially, it may reduce the risk of osteoporosis, mood swings, irritability, and other symptoms associated with fluctuations in hormone levels. Additionally, research suggests that supplementing dioscin may help protect the stomach from gastric mucosal damage by inhibiting the activity of certain enzymes.
Due to the COVID-19 pandemic, the global dioscin market was estimated at $99.50 million in 2022, projected to grow to $142.30 million by 2030, with a compound annual growth rate of 6.1% from 2022 to 2030. According to the "Newswire on the Dioscin Market Size by 2030", dioscin accounts for 60% of the world's steroid product market among all steroid drug precursors. China is the largest consumer market for dioscin globally, with a market share of approximately 60%.
Yu et al. studied the subchronic toxicity of dioscin in rats, giving dioscin at a dose of 300 mg/kg for 90 days, and found that the levels of alanine aminotransferase in male and female rats significantly increased, indicating liver damage, with a dose-dependent trend in liver injury in male rats. In this case, the hepatotoxicity of dioscin may be dose-related. Therefore, further research is necessary to investigate the possibility of dioscin accumulation causing liver damage with long-term use. Based on the results of the above studies, the single compound of dioscin has hepatotoxicity, but the hepatotoxicity of traditional Chinese medicine preparations containing dioscin has not been clinically reported, which may be due to the synergistic effects of multiple components in traditional Chinese medicine.
Melo et al. conducted toxicity tests on dioscin in liver cells and V79 cells, believing that compared with the toxic side effects of dioscin on tissues, its medicinal value is more prominent. Guo et al. (2015) studied the subchronic toxicity of dioscin in mice in related toxicity tests and found that the dose level of dioscin that did not produce adverse reactions was higher than 400 mg/kg, indicating that oral administration of dioscin is relatively safe.
Dioscin is derived from plants of the Dioscoreaceae (such as Dioscorea nipponica Makino), Liliaceae, Asparagaceae (such as Ophiopogon japonicus), Zygophyllaceae (such as Tribulus terrestris), and Solanaceae (such as Solanum melongena). Among them, plants of the Dioscorea genus are known for their abundant dioscin and diosgenin content, such as D. nipponica, D. panthaica Prain et Burk.
Diosgenin (C27H42O3) is a natural steroidal saponin belonging to the spirostanol class. Its chemical name is (3β, 25R)-spirost-5-en-3-ol-3, β-ol, which is widely present in plants of the Dioscorea genus and the Fabaceae family. Diosgenin is a hydrolysis product of dioscin and can be obtained from the rhizomes of plants of the Dioscorea genus by hydrolysis, fermentation, and extraction. In addition, diosgenin is also found in some plants outside the Dioscoreaceae family, such as Tribulus terrestris in the Zygophyllaceae family and Trigonella foenum-graecum in the Fabaceae family.

Through this article, we can clearly understand the difference between dioscin and diosgenin. Although they are very similar in name, there are significant differences in chemical structure and function. Dioscin is a natural product with multiple biological activities, while diosgenin is the metabolic product of dioscin, which may have different biological activities. Understanding the difference between these two is crucial for correctly understanding their roles and applications. Hopefully, this article provides readers with a clear explanation to help them better understand and differentiate between dioscin and diosgenin, thus maximizing their potential.
[1] Li X, Liu S, Qu L, et al. Dioscin and diosgenin: Insights into their potential protective effects in cardiac diseases. Journal of Ethnopharmacology, 2021, 274: 114018.
[2] Wang D, Wang X. Diosgenin and its analogs: Potential protective agents against atherosclerosis. Drug Design, Development and Therapy, 2022: 2305-2323.
[3] Tak Y, Kaur M, Chitranashi A, et al. Fenugreek derived diosgenin as an emerging source for diabetic therapy. Frontiers in Nutrition, 2024, 11: 1280100.
 |
 |
 |