-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

Diphenic acid is an important organic compound, and its formation mechanism is a topic of significant research interest in the field of chemistry. The synthesis of diphenic acid involves various reactions and molecular interactions. A deeper understanding of its formation mechanism not only helps us comprehend the synthesis patterns of organic compounds but also provides valuable references for applications and developments in related fields. This article explores the formation mechanism of diphenic acid, analyzing its key steps and reaction pathways to provide readers with a comprehensive understanding of the compound's formation process.
Diphenic acid, an organic compound, consists of two carboxyl groups combined into a central diphenyl nucleus. It serves as a pharmaceutical intermediate and can be utilized in synthesizing certain dyes. Diphenic acid is therapeutically characterized by its potential to mitigate the risks of chronic diseases. Derivatives of diphenic acid have garnered significant attention, often found in fruits like raspberries, strawberries, and blackberries.
Mechanism refers to the chemical processes involved in its formation and reactions. Understanding this mechanism is crucial for various reasons. It can reveal how diphenic acid is produced in natural environments, such as through microbial degradation of naphthalene. Additionally, understanding its mechanism can guide the design of new synthetic methods for producing diphenic acid or its derivatives, potentially applicable in materials science or other fields.
Diphenic acid is an organic compound centered around a diphenyl nucleus. Here's a breakdown of its primary structural components:

This formula reveals the exact number of each element in a diphenic acid molecule: 14 carbon (C) atoms, 10 hydrogen (H) atoms, and 4 oxygen atoms.
Imagine two benzene rings (alternating single and double bonds forming six-membered carbon rings) directly linked to each other. This combination forms the diphenyl nucleus.
Attached to each carbon atom on the outer layer (ortho position) of the diphenyl nucleus is a carboxyl group (COOH). Each carboxyl group consists of a carbonyl group (C=O) bonded to an oxygen atom (O), which, in turn, is connected to a hydroxyl group (OH). Essentially, there's a carboxyl group at each end of the two benzene rings.
The condensed structural formula of diphenic acid is (C6H4CO2H)2. Diphenic acid is a white solid at room temperature. It exhibits a property known as anti-isomerism, meaning molecules can exist in two non-mirror-image forms due to restricted rotation around the carbon-carbon bond connecting the two benzene rings.
There are two primary mechanisms for the preparation of diphenic acid:

This method involves converting ortho-aminobenzoic acid to its diazonium salt, followed by reduction to diphenic acid using copper(I) as a catalyst. The specific steps are as follows:
50 grams (0.365 moles) of ortho-aminobenzoic acid (m.p. 143-144s) are ground in a mortar with 150 mL of water and 92 mL of concentrated hydrochloric acid (sp. gr. 1.19). The suspension is transferred to a 1-liter round-bottom flask fitted with a mechanical stirrer. The flask is surrounded by an ice bath. When the contents cool to 0-5°C, a solution of 26.3 g (0.38 moles) of sodium nitrite dissolved in 350 mL of water is added dropwise from a dropping funnel within 30 minutes. The resulting diazonium solution is kept below 5°C and immediately filtered through a cooled Büchner funnel before use.
126 grams (0.505 moles) of copper sulfate pentahydrate are dissolved in 500 mL of water in a 2-liter beaker, and 210 mL of concentrated ammonium hydroxide (sp. gr. 0.90) are added. The solution is then cooled to 10°C. A solution of 42 g (0.256 moles) of commercial hydroxylamine sulfate (Note 1) in 120 mL of water is prepared and cooled to 10°C; 85 mL of 6N sodium hydroxide solution is added to the latter solution, and if not clear, the resulting hydroxylamine solution is clarified by suction filtration. The hydroxylamine solution is immediately added to the copper sulfate solution with hand stirring. Effervescence occurs immediately; gas is evolved, and the solution turns pale blue. If this solution cannot be used immediately, it should be protected from the air.
The reduction solution in a 2-liter beaker is surrounded by an ice bath to maintain the solution temperature at around 10°C. The beaker is equipped with a mechanical stirrer. A glass tube is mounted on the rod of a 100 mL cylindrical dropping funnel, which tube dips below the surface of the solution and is bent upward and drawn in to give an opening of about 2 mm. 80 to 90 mL of diazonium solution is placed in the dropping funnel and added at a rate of about 10 mL per minute. The remaining portion of the diazonium solution is added at the same rate, and stirring is continued for five minutes.
The solution is then rapidly heated to boiling and cautiously acidified with 250 mL of concentrated hydrochloric acid. Diphenic acid precipitates as light brown crystals. After the solution stands overnight, the solid is removed by filtration, and the mother liquor is washed with about 50 mL of cold water on the filter. After drying, the product melts at 222-227°C, weighs 39-40 g (88-91% of theory).
The crude product is suspended in 200 mL of water, and 40 g of solid sodium bicarbonate is added. The resulting solution is filtered by gravity, and the filtrate is boiled with 0.1 g of Norite. The mixture is filtered, and the yellow-brown filtrate is acidified with excess 6N hydrochloric acid while still hot. The precipitated diphenic acid is collected on the filter, and washed with 40 mL of cold water. When dried at 100°C, the product melts at 225-228°C, weighs 32-37 g (72-84% of theory). It is a light cream color.
Biphenyl can be oxidized by various oxidants such as peracetic acid, hydrogen peroxide, chromium trioxide, potassium dichromate, or potassium permanganate. This process yields biphenyl quinone as an intermediate, which further oxidizes to diphenic acid.
Designing new catalysts to improve the efficiency and selectivity of diphenic acid synthesis. This includes exploring new catalyst materials, understanding the interaction of catalysts with substrates, and optimizing reaction conditions.
Investigating alternative activation methods such as microwave or ultrasound irradiation. These techniques have the potential to accelerate reactions, increase yields, or make reaction conditions milder.
Exploring the possibility of enzymatic catalysis for diphenic acid formation. This could provide a more sustainable and environmentally friendly approach compared to traditional chemical methods.
These techniques will allow researchers to monitor reactions in real-time, providing valuable insights into reaction kinetics and enabling better process control.
Utilizing machine learning algorithms to analyze large datasets of reaction data. This can help identify patterns, predict reaction outcomes, and guide the development of new synthetic strategies.
By exploring these innovations, scientists can gain a deeper understanding of the mechanism of diphenic acid and develop more efficient, selective, and sustainable production methods. This will open doors to new applications of diphenic acid across various fields.
Diphenic acid serves as a precursor for certain dyes. Dyes, colored compounds, are used for coloring textiles, paper, plastics, and other materials.
Researchers are intrigued by diphenic acid derivatives due to their potential therapeutic properties. These derivatives may offer health benefits, such as reducing the risk of chronic diseases like cancer and cardiovascular diseases. However, further research is needed to substantiate these potential benefits. It is important to note that diphenic acid itself is not a drug and should not be consumed for any medical condition.
Research has designed the synthesis of an anion 124 fluorescent receptor based on the diphenyl motif. Initially, the diol chloride 119 reacted with 3-aminopyridine to yield the diamine 122. The diamine 122 refluxed in the presence of 9-chloromethyl anthracene to obtain the chloride salt 123, which was exchanged with NH4PF6 anion to yield the receptor 124 as a pale yellow solid.

Costero et al. synthesized a novel multinuclear azine with a 4,4'-substituted diphenyl segment, clearly indicating that modification of the dihedral angle between the diphenyl rings is a crucial factor determining the molecular fluorescence response. Fluorescence, pH-dependent due to protonated aliphatic nitrogen and carbonyl oxygen forming intramolecular hydrogen bonds, was achieved. Ligands were prepared from 4,4′-dinitro-2,2′-diphenic acid chloride 125 and the corresponding diamines 126a-c, which reacted with formaldehyde under reducing conditions (H2, Pd(C)) to yield compounds 128a-c, respectively.

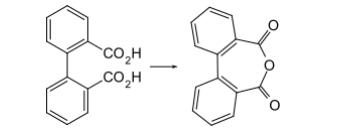
Hyperbranched polymers, including highly branched macromolecules, are garnering increasing attention due to their unique structures, diverse properties, and wide-ranging application prospects. Aromatic and semi-aromatic hyperbranched poly(ester-amides) 135 and 136 were synthesized via the reaction of diphenic anhydride 24 with polyhydroxy amines 133 and 134.

Furthermore, a two-dimensional (2D) coordination polymer [Zn-(ATZ)2]n (HATZ = 5-amino-1htetrazole) with a 2D + 2D→2D columnar layer array was synthesized. The 2,2'-diphenic acid bridging ligand connected two honeycomb-shaped Zn(ATZ)1.5 sublayers via bridging ligands of different lengths and types, forming four three-dimensional (3D) isostructural frameworks under solvothermal conditions.
Diphenic acid 1 reacted with R3Al (R = Et, i-Bu) in a 1:2 molar ratio to produce dialkylaluminum dicarboxylate salts [Et4Al2(OOCC12H8COO)]3 (137) and [i-Bu4Al2-(OOCC12H8COO)]2 (138), with yields of 55% and 40%, respectively.

In summary, the formation mechanism of diphenic acid is a complex and intricate process involving various chemical reactions and intermolecular interactions. Through the introduction in this article, we have gained a deeper understanding of the formation mechanism of diphenic acid, aiding in better grasping the synthetic pathways and reaction patterns of this important organic compound. As scientific research continues to advance and technology develops, we believe there are still many unexplored areas waiting for us to explore regarding the formation mechanism of diphenic acid.
[1] Salem M A, Helel M H, Ammar Y A, et al. Diphenic acid derivatives: Synthesis, reactions, and applications[J]. Synthetic Communications, 2017, 47(10): 935-960.
[2] https://pubchem.ncbi.nlm.nih.gov/compound/10210
[3] https://en.wikipedia.org/wiki/Diphenic_acid
[4] https://orgsyn.org/demo.aspx?prep=cv1p0222
[5] DIPHENIC ACID. Organic Syntheses. 7: 30. 1927. doi:10.15227/orgsyn.007.0030.
 |
 |
 |