-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

Understanding Diphenic Acid and Diphenic Acid Anhydride: Diphenic acid is a compound derived from biphenyl, a hydrocarbon compound naturally found in coal tar and petroleum. It holds significance in various industrial applications. Diphenic acid anhydride is a key component in the production of certain resins and advanced polymers. Diphenic acid can be synthesized from biphenyl through oxidation with peracetic acid. Microbial action can also convert biphenyl to dibenzoic acid, which can undergo intramolecular rearrangement to form the cyclic anhydride structure of dibenzoic acid anhydride. This transformation process provides a potential biological pathway for producing this valuable industrial chemical.
Diphenic acid, also known as dibenzoic acid, is an organic compound with the chemical formula (C6H4CO2H)2. It is one of several isomers of dibenzoic acids extensively studied. It is a white solid that can be prepared in the laboratory by diazotization of anthranilic acid. It forms various coordination polymers and exhibits tautomerism, forming an internal anhydride characterized by a seven-membered ring fused with two phenyl rings.
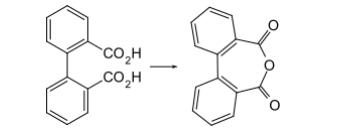
Diphenic acid anhydride, also known as dibenzofuran-5,7-dione or 2,2'-diphenic acid anhydride, is an organic compound with the chemical formula C14H8O3. It is a white solid sparingly soluble in water. It is used as a chemical intermediate in the production of dyes, pigments, and pharmaceuticals. Diphenic anhydride is a reactive compound capable of undergoing various reactions, acting as an acidic anhydride that can react with water to yield diphenic acid and with alcohols, amines, and other nucleophilic reagents.
Diphenic acid and diphenic acid anhydride are closely related compounds. Diphenic acid can undergo a dehydration process to form diphenic acid anhydride.
Benzoic acid (also known as diphenic acid) is a dicarboxylic acid, meaning it has two carboxyl groups (COOH) attached to a central benzene ring. Diphenic acid anhydride is an acid anhydride formed by the condensation of two molecules of benzoic acid with the elimination of one water molecule.
Taking diphenic acid anhydride as an example, the two carboxyl groups of diphenic acid react with each other, eliminating a water molecule to form the cyclic anhydride structure:
2 C6H5COOH (diphenic acid) → C6H5-C(O)-O-C(O)-C6H5 (diphenic acid anhydride) + H2O (water)
In essence, diphenic acid loses a water molecule to become diphenic acid anhydride.
Diphenic acid is a compound derived from biphenyl, a hydrocarbon compound naturally found in coal tar and petroleum.
Diphenic acid can be synthesized from biphenyl through oxidation with peracetic acid. Peracetic acid is prepared first from acetic acid and 90% hydrogen peroxide:
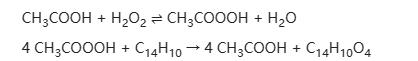
This is a mature method utilizing peracetic acid generated in situ from acetic acid and hydrogen peroxide as the oxidant. The reaction typically takes place in acetic acid, with the temperature raised to around 80-85°C. Biphenyl is first oxidized to biphenyl quinone and then further oxidized to diphenic acid. That is, biphenyl + peracetic acid → diphenic acid + acetic acid
While peracetic acid is a common choice, biphenyl can also be treated with other oxidants such as hydrogen peroxide, chromium trioxide, potassium chromate, or potassium permanganate, first producing biphenyl quinone, which is further oxidized to diphenic acid. Biphenyl quinone can be boiled in alcoholic potassium hydroxide to yield diphenic acid potassium salt or photooxidized to diphenic acid.
Ozonolysis is another method to convert biphenyl to diphenic acid. This process involves the reaction of biphenyl with ozone, which cleaves the double bonds in the molecule. The resulting products are further processed to obtain diphenic acid. Biphenyl rapidly reacts with one mole of ozone at the 9,10-double bond. Polyozonide (II) is obtained in chloroform, which can be converted to diphenic acid. While this method offers good yields, it may not be as practical due to the specialized equipment needed to safely handle ozone.
Diphenic acid serves as a valuable intermediate in synthesizing certain dyes. Dyes, colored compounds used for coloring textiles, plastics, and other materials, are dependent on specific reactions and other starting materials for production.
Interest surrounds diphenic acid derivatives due to their potential therapeutic properties. These derivatives may hold promise in reducing the risk of chronic diseases such as cancer, cardiovascular ailments, and other pathologies. However, it is crucial to note that these findings are preliminary and necessitate further research to ascertain the safety and efficacy of diphenic acid derivatives in these applications.
The rigid structure of diphenic acid, characterized by the "∞" (infinity) shape formed by two connected benzene rings, confers value in the realm of bioorganic chemistry through "conformational locking." Incorporating diphenic acid into molecules can restrict their flexibility and enforce specific conformations vital for desired functionalities. For instance, research utilizes diphenic acid to create double helical structures with explicit chirality, potentially beneficial for drug development and molecular recognition studies.
While diphenic acid anhydride is associated with diphenic acid, it lacks widely documented benefits or advantages. Here's what we know:
Unlike diphenic acid, diphenic acid anhydride appears to lack established industrial uses. Research on this particular compound is less common.
As a derivative of diphenic acid, it may possess some useful reactivity properties in certain chemical reactions. In many cases, diphenic acid itself can be directly utilized.
Diphenic acid anhydride, like many anhydrides, is a reactive irritant. Contact may lead to skin and respiratory irritation.
[1] https://en.wikipedia.org/wiki/Diphenic_acid
[2] https://onlinelibrary.wiley.com/doi/10.1002/recl.19590780306
[3] https://linkinghub.elsevier.com/retrieve/pii/S0006291X98982286
[4] https://www.sarex.com/fine/product/key-products/diphenic-acid-biphenyl-22dicarboxylic-acid
 |
 |
 |