-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

Diphenic acid is a significant organic compound with various applications and research value. Its chemical structure, comprising benzene rings and carboxylic acid groups, lends itself to diverse uses in the chemical and biological domains. This article will delve into the properties, synthesis methods, application fields, and importance of diphenic acid in scientific research and industrial production.
Diphenic acid, also known as diphenylacetic acid, is an organic compound with the chemical formula (C6H4CO2H)2. It is among the most studied isomers of diphenyl-derived dicarboxylic acids. This white solid holds potential significance across various industries. Its structure consists of two phenylacetic acid groups connected by a central diphenyl core. These phenylacetic acid groups essentially comprise benzene rings with carboxylic acid (COOH) functional groups, with the diphenyl core linking two connected benzene rings. Understanding this structure is crucial for further exploring its potential applications.
Diphenic acid, with the chemical formula (C6H4CO2H)2, consists of two benzene rings linked by a single carbon-carbon bond. Each benzene ring is attached to a carboxyl group (COOH) at the ortho position. The structure of diphenic acid is as follows:

Diphenic acid features a central diphenyl core where two benzene rings are connected by a single carbon-carbon bond. Each benzene ring is linked to a carboxyl group (COOH) at the ortho position, imparting diphenic acid with two carboxylic acid groups, making it a dicarboxylic acid.
Diphenic acid can form various coordination polymers. It also exhibits anti-isomerization, a stereochemical phenomenon resulting from restricted rotation of single bonds.
Diphenic acid is employed in dye synthesis. Diphenic acid derivatives are garnering widespread attention due to their potential therapeutic effects. Many diphenic acid derivatives have the potential to reduce the risk of chronic diseases such as cancer, cardiovascular diseases, and other pathologies. It finds applications in numerous herbal and food ingredients.
Diphenic acid esters (polyphenols) and their ellagic acid derivatives are extracted from the dried fruits of Terminalia bellerica (Bahera) and reportedly possess analgesic activity. Diphenic acid derivatives are commonly found in fruits like blackberries, raspberries, and strawberries. Consumption of these fruits has been shown to reduce the risk of chronic diseases such as cancer, cardiovascular diseases, and other ailments.
Diphenic acid is a multifunctional compound with various potential applications. As research progresses, we may see further utilization of this compound.
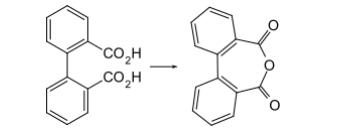
It can be produced in laboratory environments through two common methods.

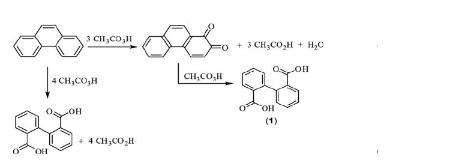
Other oxidizing agents such as chromium trioxide or potassium permanganate can also be employed for this purpose, but they typically involve multiple steps, initially converting phthalic acid to phthalic anhydride before further conversion to diphenic acid.
Diphenic acid is a gray-white solid.
Two reported values exist for the melting point of diphenic acid: 235.5°C (Wikipedia) and 227-229°C (literature value, based on previously published scientific findings). The difference between these values is relatively small, possibly due to variations in sample purity or measurement techniques. It can be concluded that the melting point of diphenic acid is around 230°C.
Diphenic acid can be purchased from chemical suppliers, typically offering high-purity grades. It should be noted that if mishandled, diphenic acid can be a hazardous substance. Observing safety protocols during handling is crucial.
Current research on diphenic acid primarily focuses on:
Through this introduction, we've gained a deeper understanding of diphenic acid's structure, properties, synthesis methods, and applications. As scientific and technological advancements continue, research and applications of diphenic acid will expand, bringing forth new possibilities and opportunities across various domains. It is hoped that this article provides valuable information to readers and inspires further exploration and application of diphenic acid.
[1] https://en.wikipedia.org/wiki/Diphenic_acid
[2] https://www.sarex.com/fine/product/key-products/diphenic-acid-biphenyl-22dicarboxylic-acid
[3] https://www.citychemical.com/diphenic-acid.html
[4] Salem M A, Helel M H, Ammar Y A, et al. Diphenic acid derivatives: Synthesis, reactions, and applications[J]. Synthetic Communications, 2017, 47(10): 935-960.
 |
 |
 |