-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

Ethyl cinnamate, an organic compound with the chemical formula C11H12O2, is an ester substance formed by the esterification reaction of cinnamic acid and ethanol. It possesses a sweet aroma reminiscent of a blend of cinnamon and fruit, making it widely used in food flavorings, perfumes, and cosmetics. Besides its extensive application in the flavor and fragrance industry, ethyl cinnamate also serves significant roles in pharmaceuticals and organic synthesis as an intermediate for various compounds. This article provides a detailed overview of the chemical properties, structure, and applications of ethyl cinnamate across various fields, aiding in a better understa nding of this important chemical substance.
Ethyl cinnamate, also known as β-phenylacrylic acid ethyl ester, can be extracted from plant sources like cinnamon leaf oil. It is an important fragrance ingredient used in oriental and fruity fragrances, with widespread applications in daily chemicals, food, and more.
Ethyl cinnamate is an organic compound found in cinnamon and other plants. It is a colorless to pale yellow liquid with a pleasant fruity-cinnamon odor. Here are some properties of ethyl cinnamate:
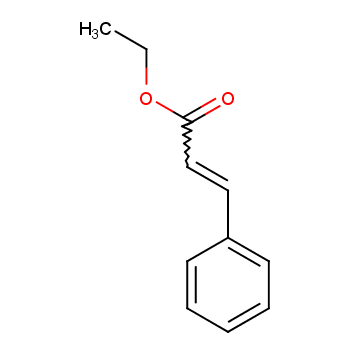
(1) Ethyl cinnamate Chemical formula: C11H12O2
(2) Ethyl cinnamate Molar mass: 176.21 g/mol
(3) Ethyl cinnamate Density: 1.05 g/cm3
(4) Ethyl cinnamate Melting point: 6.5 to 8°C (43.7 to 46.4°F)
(5) Ethyl cinnamate Boiling point: 271°C (519.8°F)
(6) Ethyl cinnamate Refractive index: n20/D 1.558 (lit.)
(7) Ethyl cinnamate Solubility: Soluble in chloroform, dichloromethane, ethyl acetate, DMSO, acetone, etc.
The molecular formula of ethyl cinnamate is C11H12O2. How do we analyze ethyl cinnamate structure? The specific analytical approach is illustrated in the diagram below. First, determine the unsaturation of ethyl cinnamate as 5, and the possible functional groups in the structure may include hydroxyl, carboxyl, ester, and carbon-carbon double bonds. Then, infer the types of carbon-carbon bonds and functional groups based on unsaturation. Subsequently, use physical and chemical methods to determine the types and quantities of functional groups in ethyl cinnamate, where physical methods mainly involve instrumental spectroscopic analysis and chemical methods involve property experiments.
Analyzing the ethyl cinnamate ir spectrum, as shown in the table below, reveals the presence of ester groups, benzene rings, and carbon-carbon double bonds.

The hydrogen spectrum of ethyl cinnamate shows six chemical environments of hydrogen atoms, with peak areas in a ratio of 3:2:1:3:2:1 from left to right. As a mono-substituted compound, due to the electron-withdrawing and conjugation effects of the substituent, only two types of hydrogen atoms exist on the benzene ring.

Based on the available information, the structural formula of ethyl cinnamate can be deduced.

Ethyl cinnamate finds applications in various fields, including:
(1) Fragrances: It is a common fragrance component used in creating fruity, spicy, and incense-like scents.
(2) Food flavorings: Used in candies, chewing gum, beverages, and more to impart a cinnamon flavor.
(3) Pharmaceuticals: It exhibits some antifungal and antibacterial properties, sometimes used in topical medications.
It is noteworthy that ethyl cinnamate may be a skin irritant for some individuals. It is also a potential allergen.
Currently, the main synthetic route for ethyl cinnamate involves the synthesis of cinnamic acid and ethanol catalyzed by a catalyst. In an industrialized process, concentrated sulfuric acid is used as a catalyst, but this process suffers from low product yield, severe equipment corrosion, low product selectivity, and large wastewater volume. Some researchers have used cation exchange resin as a catalyst for the synthesis of ethyl cinnamate, which has advantages such as low catalyst cost, simple and safe synthesis process, and high catalytic activity, but the product yield is relatively low. Li Jing et al. synthesized ethyl cinnamate using heteropoly acid as a catalyst and carried out the reaction under microwave assistance, using molecular sieve as a dehydrating agent, achieving a total yield of 95%, but the high synthesis cost limits industrialization. Ding Liang et al. used iron chloride as a catalyst loaded on polyvinyl chloride resin to obtain ethyl cinnamate, realizing a synthetic route catalyzed by inorganic salt, but the yield was only 80%. Yu Shanxin et al. synthesized ethyl cinnamate using ammonium iron sulfate as a catalyst with a yield of 94%, but industrialization was not achieved.
Hao Lina et al. used cinnamic acid and ethanol as raw materials, and calcium sulfate hemihydrate as a catalyst to synthesize ethyl cinnamate. Based on single-factor experiments, the Box-Behnken response surface method was used to optimize the synthesis process. The optimal process conditions were found to be: the molar ratio of cinnamic acid to ethanol was 1:6.36, calcium sulfate hemihydrate addition was 12.86%, reaction time was 14.55 hours, and the yield of ethyl cinnamate could reach 89.42%. With low cost and suitability for industrial production.
Ethyl cinnamate provides a complex aroma. It has a familiar cinnamon scent along with fruity and spicy notes. Imagine a sweet and warm aroma of cinnamon intertwined with hints of honey, red berries, and vanilla. This unique fragrance makes ethyl cinnamate a popular choice in perfumes and flavorings.
In its purest form, ethyl cinnamate is a colorless to pale yellow liquid. However, commercially available ethyl cinnamate may sometimes appear as a solid, depending on storage conditions. It's worth noting that this does not affect its properties, and it easily liquefies upon slight heating.
Ethyl cinnamate, with its unique structure, excellent chemical properties, and diverse synthesis methods, has become an important compound in multiple industries. Its sweet aroma finds widespread applications in food flavorings, perfumes, and cosmetics, while its stability and bioactivity make it valuable in pharmaceuticals and organic synthesis. With ongoing technological advancements and deeper research into ethyl cinnamate, the prospects for its applications are broadening, bringing forth more innovation and development opportunities for related industries. Through further optimization of synthesis processes and expansion of application areas, ethyl cinnamate is poised to play a greater role in the future.
[1] Xie, D., Zheng, L., & Liu, C. (2024). Design of theme-based review teaching of "derivatives of hydrocarbons" based on real situations — Understanding ethyl cinnamate. Chemical Education, 45(09), 54-60. DOI:10.13884/j.1003-3807hxjy.2023030021.
[2] Hao, L., & Zhang, L. (2019). Research on the synthesis of ethyl cinnamate catalyzed by calcium sulfate hemihydrate. Contemporary Chemical Industry, 48(10), 2223-2226. DOI:10.13840/j.cnki.cn21-1457/tq.2019.10.012.
[3]https://pubchem.ncbi.nlm.nih.gov/compound/Ethyl-cinnamate
 |
 |
 |