Fludarabine is a purine nucleoside analog commonly used to treat chronic lymphocytic leukemia (CLL) and other types of lymphomas. Compared to other chemotherapy drugs, fludarabine has relatively low toxicity but can still cause some serious side effects.
Fludarabine is a nucleoside analog and belongs to the purine analog class of anticancer drugs. It exerts cytotoxic effects by interfering with DNA synthesis, inhibiting RNA polymerase II activity, and inducing apoptosis in various tumor cells. Approved by the FDA in 1984, fludarabine was initially used to treat CLL. It is now employed in the management of other hematologic malignancies such as follicular lymphoma, small lymphocytic lymphoma, and mantle cell lymphoma. The primary fludarabine toxicity is bone marrow suppression, leading to leukopenia, neutropenia, and anemia. Fludarabine can also cause various neurological toxicities, ranging from mild peripheral neuropathy to severe central nervous system manifestations such as hallucinations, motor weakness, paralysis, or seizures.
Although uncommon at recommended doses, acute fludarabine toxicity can manifest as severe neurological effects, including blindness, coma, and seizures, which may appear weeks after treatment initiation. Immediate medical attention is crucial in such cases, with treatment strategies focusing on symptomatic supportive care and potential anti-inflammatory medication to alleviate symptoms.
What are the long term effects of fludarabine? Common side effects include nausea, diarrhea, fever, rash, dyspnea, numbness, visual changes, and fatigue. Severe adverse effects comprise cerebral dysfunction, low blood cell counts, and pulmonary inflammation. Fludarabine use during pregnancy may cause harm to the fetus. Prolonged fludarabine administration is more prone to induce chronic adverse effects, including bone marrow suppression leading to decreased red blood cells, white blood cells, and platelets, increasing the risk of infections, anemia, and bleeding. Regular monitoring of blood cell counts during treatment is essential for early detection of these effects.
Does fludarabine cause neuropathy? Fludarabine, a nucleoside analog with immunosuppressive properties, exhibits synergistic effects with alkylating agents. High-dose fludarabine in early dose-finding studies reported dose-dependent severe central nervous system toxicity. Notably, high doses of fludarabine (>96 mg/m^2/day for ≥ 5-7 days, approximately four times the recommended dose) led to a syndrome characterized by blindness, progressive encephalopathy, or even death in 36% of patients. Lower doses of fludarabine are associated with a reduced incidence of neurological toxicity. Nonetheless, reports of neurological toxicity persist even with standard doses of fludarabine, especially in low-intensity preconditioning regimens.
In a retrospective institutional review, Beitinjaneh et al. reported severe fludarabine-related leukoencephalopathy in 39 out of 1596 adult and pediatric patients undergoing hematopoietic stem cell transplantation (HSCT). Three clinical syndromes were described: posterior reversible encephalopathy syndrome (PRES) characterized by seizures, headaches, visual changes, and variable mental status alterations with cortical and subcortical white matter involvement on brain MRI; acute toxic leukoencephalopathy (ATL) featuring cognitive impairment, decreased consciousness, visual changes, and deep white matter changes on MRI; and other ATL-like deep white matter leukoencephalopathy with less prominent MRI white matter changes. Central nervous system toxicity typically occurs approximately two months after initiating fludarabine and tends to stabilize in severity over the following month. Of the 14 patients (36%) who experienced fludarabine-related neurotoxicity, all deaths occurred within six months post-toxicity onset. Among the 10 patients with fludarabine-related neurotoxicity who survived HSCT for over a year and remained non-lost to follow-up, neurological recovery was observed in six patients, three had partial recovery, and one had persistent neurological deficits.
Is fludarabine cardiotoxic? The most common adverse effects of fludarabine are bone marrow suppression and immunosuppression. Although cardiac complications with fludarabine are rare, there have been reports of congestive heart failure or left ventricular dysfunction. The most common adverse effects of anticancer chemotherapy drugs (CCAs) are nausea, vomiting, alopecia, stomatitis, and leukopenia. Two CCAs associated with cardiac toxicity are mitoxantrone and fludarabine. Mitoxantrone, an anthracycline, has been reported to cause ventricular arrhythmias. Fludarabine, a purine analog, is rarely associated with cardiac dysfunction. These therapies have been co-administered in the treatment of multiple myeloma, CLL, chronic myeloid leukemia, acute lymphoblastic leukemia, non-Hodgkin lymphoma, acute myeloid leukemia, and paroxysmal nocturnal hemoglobinuria, with good tolerability. While these drugs individually do not typically cause cardiac toxicity, the risk increases when used in combination, leading to a greater potential for congestive heart failure.
In November 2005, a 22-year-old male diagnosed with acute myeloid leukemia achieved complete remission after two courses of idarubicin induction chemotherapy (12 mg/m^2 intravenous infusion for 3 days) and cytarabine (200 mg/m^2 intravenous infusion for 7 days). Subsequently, he received two additional courses of cytarabine (3000 mg/m^2)2 intravenous infusion over 3 hours, twice daily, for 3 days each, for consolidation in April 2006. Six months later, leukemia relapsed, unresponsive to re-induction chemotherapy (idarubicin 12 mg/m^2 intravenously on days 1-3 and cytarabine 200 mg/m^2 on days 1-7). Aside from bone marrow suppression, no toxic events occurred during idarubicin and cytarabine treatment. Given the refractoriness of re-induction chemotherapy, salvage chemotherapy with the FLAGI regimen (fludarabine 30 mg/m^2 intravenously on days 1-5, cytarabine 2000 mg/m^2 on days 1-5, and idarubicin 12 mg/m^2 on days 7-8) was prescribed. His electrocardiogram revealed:
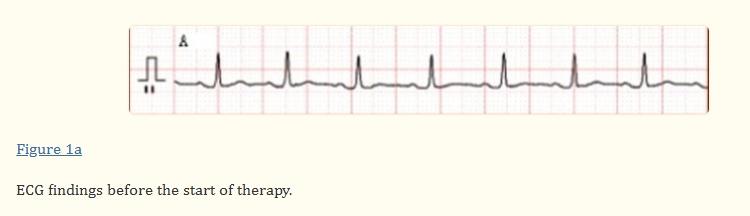
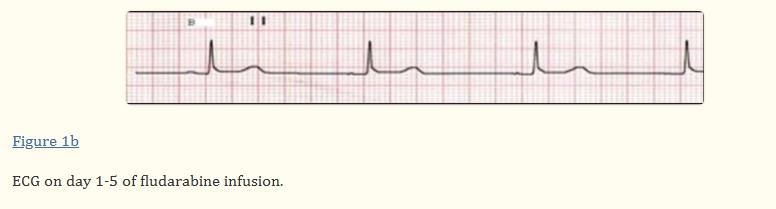
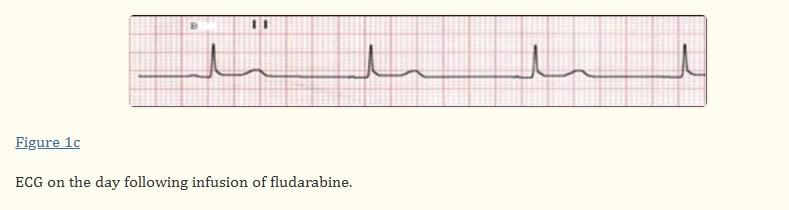
The patient's bradycardia had multiple potential causes. Sepsis, cardiac events, electrolyte imbalances, and cytarabine, among other drugs, could lead to bradycardia. Drug adverse reactions were considered a likely cause of bradycardia in this patient, as no other obvious causes were identified. Bradycardia occurred during fludarabine treatment and markedly subsided after completing the drug infusion. Other potential causes of bradycardia were ruled out, thus suggesting a plausible association between fludarabine and sinus bradycardia observed in this patient. Clinicians should be aware of this potential fludarabine toxicity, particularly considering its increased use in treating hematologic diseases.
(1) Regular follow-up visits with your doctor to monitor your progress and ensure the medication's efficacy are crucial. Blood tests may be necessary to check for adverse reactions.
(2) Refrain from any immunizations (vaccinations) without medical approval while receiving fludarabine injections and after discontinuing treatment. Fludarabine injections may lower your body's immunity, increasing the risk of infection from vaccinations. Additionally, individuals in your household should not receive oral polio vaccine as they might transmit the poliovirus to you. Avoid close contact with individuals who have recently received oral polio vaccine. If unable to take these precautions, consider wearing a protective mask covering your nose and mouth.
(3) Fludarabine injections may temporarily reduce white blood cell counts, increasing the chance of infections (e.g., pneumonia). It may also decrease platelet counts, essential for normal blood clotting. In such cases, certain preventive measures can reduce the risk of infection or bleeding, especially when blood cell counts are low:
- Avoid contact with individuals who have infections. If you suspect an infection, fever, chills, coughing, hoarseness, back or side pain, pain or difficulty urinating, consult your doctor immediately.
- Monitor for shortness of breath or abnormal bleeding or bruising.
- If you notice any abnormal bleeding or bruising, consult your doctor immediately. Watch out for black, tarry stools; blood in urine or stool; or pinpoint red spots on the skin.
- Be cautious when using a regular toothbrush, dental floss, or toothpick. Your doctor, dentist, or nurse may recommend alternative methods for cleaning teeth and gums. Consult your doctor before any dental work.
- Avoid touching the inside of your nose or eyes unless you have just washed your hands and not touched anything else during that time.
- Be careful when using a safety razor or any sharp objects such as nail or toenail clippers to avoid cutting yourself.
- Avoid contact sports or other situations where bruising or injury may occur.
- Do not take this medication if you are also taking pentostatin, as concurrent use may increase the risk of severe side effects.
(4) This medication may cause a severe reaction known as tumor lysis syndrome. Your doctor may prescribe a medication to help prevent this condition. Call your doctor immediately if you experience decreased or changing urine output; joint pain, stiffness, or swelling; lower back, side, or stomach pain; rapid weight gain; swelling of the feet or lower legs; or unusual tiredness or weakness.
(5) Using this medication during pregnancy can harm the unborn baby. Use effective birth control methods to prevent pregnancy. You should not become pregnant while taking this medication and for 6 months after stopping it. If you think you may be pregnant while using the medication, inform your doctor immediately.
(6) This medication may cause unusual weakness, difficulty thinking, or blurred vision. Before driving, operating machinery, or engaging in activities that require you to be alert, coordinated, or able to think or see clearly, make sure you know how you react to this medication.
Unfortunately, there is no specific antidote for fludarabine overdose. Immediate medical attention is crucial if you or someone you know overdoses on fludarabine.
Fludarabine can be harmful, especially at high doses. It can suppress bone marrow function, resulting in reduced counts of vital blood cells such as red and white blood cells. This increases the risk of infections, anemia, and bleeding. Other potential side effects include nausea, vomiting, and neurological issues. Due to these risks, fludarabine is prescribed only under close medical supervision and in specific circumstances.
The most common adverse reaction to fludarabine is bone marrow suppression, including leukopenia, neutropenia, and anemia. While these side effects are usually reversible, they may lead to severe infections and other complications. Other common side effects include nausea, vomiting, diarrhea, fever, and fatigue. The fludarabine toxicity depends on various factors, including the patient's age, health status, and the dose of medication received. Physicians carefully monitor patients for adverse effects during fludarabine treatment and adjust the dosage or discontinue treatment as needed. Overall, fludarabine is an effective anticancer drug, but it can still cause some serious side effects. Patients should discuss the risks and benefits of fludarabine with their doctors to determine if this medication is suitable for them.
[1]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6718929/
[2]https://www.sciencedirect.com/topics/neuroscience/fludarabine
[3]https://www.astctjournal.org/article/S1083-8791(14)01026-X/fulltext
[4]Woei C L, Ching Yun H, Chang Fang C, et al. Fludarabine-induced bradycardia in a patient with refractory leukemia[J]. 2010.
[5]https://www.mayoclinic.org/drugs-supplements/fludarabine-intravenous-route/side-effects/drg-20063840?p=1
[6]https://go.drugbank.com/drugs/DB00322
[7]https://www.fda.gov/media/163105/
 |
 |
 |