Fludarabine is a key anticancer drug widely used in the treatment of various hematologic malignancies. Fludarabine uses include treating chronic lymphocytic leukemia (CLL), non-Hodgkin lymphoma (NHL), and other lymphoproliferative disorders. As a purine nucleoside analog, fludarabine inhibits DNA synthesis and repair, halting the proliferation and growth of cancer cells, showing significant efficacy. Additionally, fludarabine is also employed in certain pretransplant conditioning regimens for both autologous and allogeneic stem cell transplantation to improve transplant success rates and reduce the risk of relapse. This article will delve into the main fludarabine uses and its clinical effectiveness in different cancer treatments.
Fludarabine is a chemotherapy drug used to treat certain types of cancer, especially in adult patients with chronic lymphocytic leukemia (CLL) who have typically received at least one other treatment without improvement. Peripheral blood smears show CLL cells as depicted in the figure below:
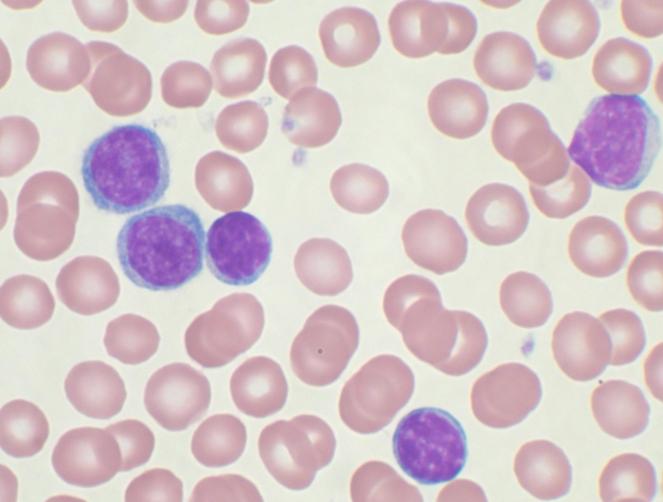
It is also sometimes used to treat non-Hodgkin lymphoma (NHL) and cutaneous T-cell lymphoma (a lymphoma affecting the skin). Formerly known by the brand name Fludara, fludarabine exerts its effects by disrupting the growth of cancer cells. It acts on DNA polymerase α, ribonucleotide reductase, and DNA primase, inhibiting DNA synthesis and disrupting cancer cells. It belongs to a class of drugs called antimetabolites, which work by mimicking substances necessary for cell growth. Once inside cancer cells, fludarabine is converted into an active form, disrupting the production of DNA and RNA necessary for cell growth and replication. Therefore, fludarabine can slow down or halt the growth of cancer cells.
Fludarabine is a purine analog and antineoplastic agent. It is typically used in its 5'-phosphorylated form, known as fludarabine phosphate, marketed under brands such as Fludara.
What is the purpose of fludarabine? Fludarabine phosphate is a purine antimetabolite approved for the treatment of patients with chronic lymphocytic leukemia (CLL). Fludarabine primarily acts by inhibiting DNA synthesis. The compound also exhibits lymphocyte toxicity, with preferential activity against T lymphocytes. Preliminary preclinical studies have shown antitumor activity of fludarabine against L1210 mouse leukemia. In Phase I studies, bone marrow suppression was identified as the dose-limiting toxicity in solid tumor patients, and fatal neurotoxicity was identified as the dose-limiting toxicity in adult acute hematologic malignancy patients. Recommended doses and schedules were determined to be 18-25 mg/m2/d, administered for five days, repeated every 28 days. In contrast to preclinical studies, Phase II trials suggested no significant effect when fludarabine was used in solid tumor patients. However, Phase II studies have confirmed the efficacy of fludarabine against malignancies of the lymphatic system (including non-Hodgkin lymphoma, cutaneous T-cell lymphoma, precursor B-cell leukemia, and chronic lymphocytic leukemia). The role of fludarabine in pediatric leukemia treatment is under investigation. Early results indicate unusual antitumor activity when the drug is used in combination chemotherapy with refractory disease patients. Fludarabine is an effective antineoplastic agent for treating lymphatic system malignancies.
Fludarabine is highly effective in the treatment of chronic lymphocytic leukemia (CLL), producing higher response rates compared to alkylating agents alone. Fludarabine, in combination with cyclophosphamide, mitoxantrone, dexamethasone, and rituximab, is used in various combinations for treating indolent non-Hodgkin lymphoma. Fludarabine, along with cytarabine and granulocyte colony-stimulating factor, is used in the FLAG or FLAMSA regimen for treating acute myeloid leukemia. Due to its immunosuppressive effects, fludarabine is also used in some preconditioning regimens before allogeneic stem cell transplantation.
FLUDARA (fludarabine) is indicated for the treatment of adult patients with B-cell chronic lymphocytic leukemia (CLL) who have not responded to or have progressed during treatment with at least one standard alkylating-agent containing regimen. The safety and efficacy of fludarabine phosphate injection (FLUDARA) have not been established in untreated or non-refractory CLL patients.
Fludarabine has been studied in patients with various lymphoproliferative malignancies. Clinical evidence from comparative studies in chronic lymphocytic leukemia (CLL) suggests that fludarabine is at least as effective as CAP (cyclophosphamide, doxorubicin, and prednisone) or CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone) in previously treated or untreated patients, and significantly more effective than chlorambucil in terms of response rate, duration of response, and survival for untreated patients. Combination therapy based on fludarabine has also yielded promising results in treating CLL patients. Furthermore, sequential therapy of fludarabine and cytarabine has shown good efficacy in treating acute leukemias, and both monotherapy and combination therapy with fludarabine have demonstrated favorable outcomes in low-grade non-Hodgkin lymphoma. Favorable cytoreductive responses have been reported in patients with lymphoplasmacytic lymphoma and a minority of patients with cutaneous T-cell lymphoma and T-cell chronic lymphocytic leukemia. Recent data also support the use of fludarabine in patients undergoing peripheral blood stem cell or bone marrow transplantation, either as part of nonmyeloablative conditioning regimens or for achieving minimal residual disease. The tolerability of fludarabine is similar to CAP, with the most common adverse events being neutropenia, thrombocytopenia, anemia, and infection. The incidence of hair loss and nausea/vomiting during fludarabine treatment appears to be lower than with CAP, although fludarabine is more prone to causing immunologic cytopenias. Fludarabine has been reported to have serious neurotoxicity, but this is primarily limited to high-dose use. Therefore, clinical experience suggests that fludarabine is an effective and well-tolerated antineoplastic agent for the second-line treatment of advanced chronic lymphocytic leukemia. Recent comparative study data also support the early use of fludarabine in treatment-naive chronic lymphocytic leukemia patients. Additionally, the results of existing studies increasingly emphasize the important role of fludarabine in the treatment of acute leukemias, low-grade NHL, and possibly other lymphoproliferative disorders, especially when used as part of combination chemotherapy.
Fludarabine can be administered via intravenous injection or oral tablets.
The recommended adult dose of fludarabine for injection is 25 mg/m2 administered intravenously over approximately 30 minutes daily for five consecutive days. Each course of treatment should begin every 28 days. Dosing may be reduced or delayed based on hematologic or nonhematologic toxicity evidence. In the event of neurotoxicity, consideration should be given to delaying or discontinuing the medication.
Many clinical settings may predispose to increased toxicity with intravenous fludarabine administration. These include advanced age, renal impairment, and bone marrow impairment. Such patients should be closely monitored for excessive toxicity, and dosage adjustments should be made accordingly.
The optimal duration of treatment has not been clearly defined. After achieving maximal response, it is recommended to administer three additional cycles of fludarabine injection, followed by discontinuation. Patients with renal impairment should be closely monitored for excessive toxicity, and dosage adjustments should be made accordingly. Intravenous fludarabine (Fludara) should not be mixed with other medications.
Before starting fludarabine and before each refill, read the Patient Information Leaflet provided by your pharmacist (if available). If you have any questions, consult your doctor or pharmacist.
Take this medication by mouth as directed by your doctor, with or without food, usually once daily for five days. This 5-day cycle can be repeated every 28 days, typically for up to 6 cycles. Swallow the tablet whole with a glass of water. Do not crush, chew, or break the tablet.
The dosage depends on your medical condition, body size, and response to treatment. Your doctor will check your blood cell counts to ensure you can proceed with the next cycle. Follow all medical/lab appointments.
Do not increase the dose or take this medication more frequently than prescribed. Your condition will not improve faster, and you may increase the risk of serious side effects.
Fludarabine is a potent chemotherapy drug with significant risk of adverse effects. It should only be used under the supervision of qualified physicians who can weigh the benefits and risks of your individual circumstances.
Bone marrow suppression (severe/ cumulative), aplastic anemia, autoimmune hemolytic anemia (fatal/severe), infections, fever, chills, gastrointestinal discomfort, malaise, central nervous system effects (such as weakness, agitation, confusion, visual disturbances, coma, peripheral neuropathy), pneumonia, pulmonary allergies (such as dyspnea, interstitial lung infiltrates), stomatitis, gastrointestinal bleeding, edema, tumor lysis syndrome, rash, hemorrhagic cystitis (rare); among others.
Do you lose hair with fludarabine? You may experience hair loss. This includes your eyelashes, eyebrows, armpits, legs, and sometimes pubic hair. Once treatment ends, your hair usually grows back, but it may be softer. It may grow back a different color or more curly than before.
Bone marrow suppression. Evaluate and monitor for hemolysis. Monitor blood (especially CBC, platelets). Use irradiated blood products if transfusion is needed. Prophylaxis may be required for tumor lysis syndrome. Renal impairment. Delay or discontinue treatment if neurotoxicity occurs. Elderly, pregnant women (Cat. D): Avoid use. Breastfeeding mothers: Not recommended.
Fludarabine demonstrates significant efficacy in treating chronic lymphocytic leukemia (CLL), non-Hodgkin lymphoma (NHL), and other lymphoproliferative disorders. Additionally, it plays a crucial role in pretransplant conditioning regimens for both autologous and allogeneic stem cell transplantation. By inhibiting cancer cell DNA synthesis and repair, fludarabine effectively halts the proliferation and spread of cancer cells. Although its use may be accompanied by some side effects, under the guidance of a physician, fludarabine provides an important treatment option for many hematologic malignancy patients, significantly improving their survival rates and quality of life. Through this article, we gain a deeper understanding of the fludarabine uses. If you or your loved ones have related conditions requiring treatment, it is advisable to consult a doctor or a professional medical team for personalized treatment plans and guidance.
[1] https://books.google.com/books
[2] https://en.wikipedia.org/wiki/Chronic_lymphocytic_leukemia
[3] https://www.cancerresearchuk.org/about-cancer/treatment/drugs/fcr
[4] https://www.webmd.com/drugs/2/drug-153310/fludarabine-oral/details
[5] https://www.cancer.gov/about-cancer/treatment/drugs/fludarabinephosphate
[6] https://pubmed.ncbi.nlm.nih.gov/17661532/
[7] https://pubmed.ncbi.nlm.nih.gov/9179529/
[8] https://pubmed.ncbi.nlm.nih.gov/7532163/
[9] https://www.empr.com/drug/fludarabine/
 |
 |
 |