-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

The combined use of fludarabine and cyclophosphamide has garnered considerable attention in the treatment of hematologic malignancies. Numerous studies have explored the efficacy and safety of these two drugs in combination, particularly in the treatment of chronic lymphocytic leukemia (CLL). Fludarabine, a purine nucleoside analog, disrupts DNA synthesis and repair, thereby inhibiting cancer cell proliferation, while cyclophosphamide, an alkylating agent, kills cancer cells by forming cross-links in DNA. The combination of these two drugs aims to capitalize on their synergistic effects to enhance treatment efficacy and response rates. This article will delve into the research findings and clinical prospects of the combined use of fludarabine and cyclophosphamide.

Fludarabine belongs to a class of drugs known as antimetabolites and is used in the treatment of a cancer called B-cell chronic lymphocytic leukemia (CLL). It is employed in CLL patients who have received prior treatment with an alkylating agent such as bendamustine but have shown inadequate response.
Fludarabine disrupts the growth of cancer cells, ultimately leading to their destruction. However, it may also affect the growth of normal cells, leading to potential side effects. Some of these side effects may be severe and must be reported to a doctor, while others may be less serious but still concerning. Certain side effects may persist even after discontinuation of fludarabine. Before starting treatment with fludarabine, you and your doctor should discuss the benefits and risks of this medication. Fludarabine is only to be used under a doctor's prescription. Oral fludarabine was withdrawn from the U.S. market in September 2011.
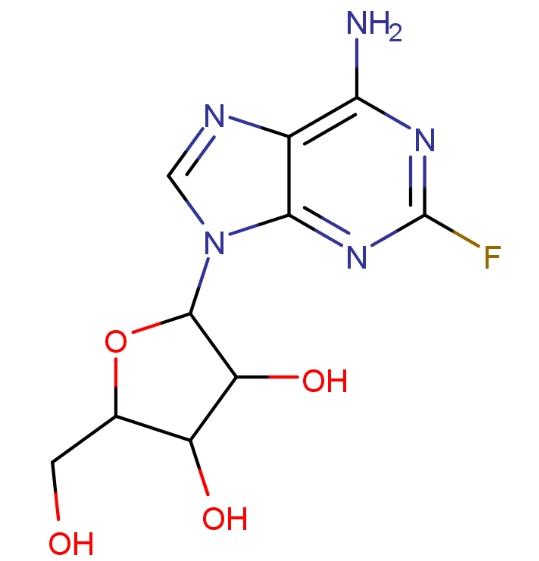
Cyclophosphamide is a precursor of alkylating agents and must be activated in the liver to form active phosphoramide. It has been used in the treatment of lymphomas and leukemia. Its side effects include hair loss, and it has been used to "de-hair" sheep. Cyclophosphamide may also lead to infertility, birth defects, mutations, and cancer.
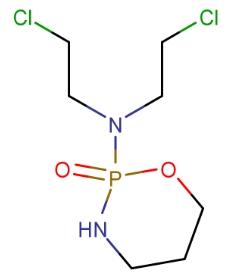
Fludarabine is a chemotherapy drug used in the treatment of chronic lymphocytic leukemia (CLL). It acts on DNA polymerase α, ribonucleotide reductase, and DNA primase, inhibiting DNA synthesis and disrupting cancer cells.
Phosphorylated fludarabine rapidly dephosphorylates to 2-fluoro-ara-A, which then undergoes intracellular phosphorylation by deoxycytidine kinase to the active triphosphate salt 2-fluoro-ara-ATP. This metabolite appears to act by inhibiting DNA polymerase α, ribonucleotide reductase, and DNA primase, thereby suppressing DNA synthesis. The exact mechanism of action of this antimetabolite is not fully understood and may involve multiple aspects.
Cyclophosphamide is an anticancer drug belonging to the class of alkylating agents, used to treat various cancers. Alkylating agents are so named because they can add alkyl groups to many electronegative groups in cells. They prevent tumor growth by cross-linking guanine bases in the DNA double helix (direct DNA attack), preventing the strands from unwinding and separating. Since this is necessary for DNA replication, cells cannot divide. Additionally, these drugs add methyl or other alkyl groups to molecules they do not belong to, which in turn inhibits their correct utilization through base pairing, leading to DNA coding errors. Alkylating agents are cell-cycle nonspecific. Alkylating agents act through three different mechanisms, all of which achieve the same end result - disruption of DNA function and cell death.
Alkylating agents act through three different mechanisms: A. Attachment of alkyl groups to DNA bases, causing DNA repair enzymes to cleave DNA when attempting to replace alkylated bases, thus preventing affected DNA from undergoing DNA synthesis and RNA transcription; B. Causing DNA damage by forming cross-links (bonds between atoms) in DNA, thereby preventing DNA separation for synthesis or transcription; C. Inducing nucleotide mispairing, leading to mutations.
Fludarabine is primarily used for treating B-cell chronic lymphocytic leukemia and non-Hodgkin's lymphoma. It is an effective and well-tolerated anticancer drug studied in various lymphoproliferative malignancies. Fludarabine is an effective treatment option for second-line therapy in advanced chronic lymphocytic leukemia. Recent comparative study data also support its early use in treating chronic lymphocytic leukemia patients who have not received prior chemotherapy. Furthermore, emerging evidence from existing studies increasingly emphasizes the significant role of fludarabine in treating acute leukemia and low-grade NHL, as well as potentially other lymphoproliferative diseases, especially when used as part of combination chemotherapy.
Intravenous cyclophosphamide injection is used to treat various malignant tumors, including Hodgkin's lymphoma, lymphoblastic lymphoma, mixed-cell lymphoma, histiocytic lymphoma, Burkitt's lymphoma, multiple myeloma, leukemia, fungal infections, neuroblastoma, ovarian adenocarcinoma, retinoblastoma, and breast cancer. Oral cyclophosphamide capsules are also used to treat pediatric nephrotic syndrome unresponsive (or intolerant) to corticosteroid therapy.
Clinical Efficacy of Fludarabine and Cyclophosphamide Combination Therapy:
FCR is an anticancer drug combination used to treat chronic lymphocytic leukemia (CLL). FCR has provided the best outcomes in terms of CR rate, PFS, and OS for newly diagnosed CLL patients to date. It comprises the drugs listed below: fludarabine, cyclophosphamide, and rituximab.
Fludarabine is a fluorinated purine nucleoside analog resistant to adenosine deaminase, initially studied as a single agent in relapsed/refractory (R/R) CLL patients, with response rates ranging from 33-57%. In treatment-naive patients, the overall response rate (ORR) to single-agent fludarabine can be as high as 70%.
To enhance the response rates of single-agent fludarabine treatment and based on in vitro evidence of synergy between fludarabine and cyclophosphamide [Yamauchi et al., 2001], CLL treatment testing of the two drugs in combination (FC) was conducted, including patients previously treated only with single-agent fludarabine. A total of 128 CLL patients received various FC regimen treatments. The overall response rate to fludarabine or alkylating agents upon study entry was greater than or equal to 80%. Further evidence of synergy between the two drugs was demonstrated, with patients resistant to fludarabine at study entry still achieving a response rate of 38% with FC combination therapy. However, the complete response (CR) rate remained low, at 35% for treatment-naive patients.
The FCR regimen was initially evaluated by the MD Anderson team in a phase II trial of R/R CLL patients. The regimen included rituximab 375 mg/m2 (day 1 of cycle 1) and 500 mg/m2 (day 1 of cycles 2-6); fludarabine 25 mg/m2/day; and cyclophosphamide 250 mg/m2/day, administered for 3 days per cycle. According to the final updated report of this trial, among 280 evaluable patients, 30% achieved CR, 14% achieved nodular partial remission (nPR), 30% achieved partial remission (PR), and the ORR was 74%. Patients who had received 3 or more prior therapies had significantly higher CR and PR rates compared to those who had received 4 or more prior therapies (CR/nPR = 52% vs. 4%, p < 0.0001). Patients previously treated with rituximab or fludarabine alone (without alkylating agents) did not have a negative impact on response rates (CR/nPR 62% and ORR 84%); however, patients treated with both fludarabine and alkylating agents exhibited moderate response rates (CR/nPR 42% and ORR 73%), with response rates significantly lower in fludarabine-resistant patients compared to those who had responded to regimens containing fludarabine previously (CR/nPR = 8% vs. 46%, p = 0.023). The median progression-free survival (PFS) for patients achieving CR was 60 months, for nPR patients it was 38 months (p = 0.076), and for PR patients it was 15 months (p < 0.001). Patients with high-risk cytogenetic abnormalities (del 17p, del 11q, 3 or more cytogenetic abnormalities) had lower PFS.
Subsequently, in a phase II clinical trial, the FCR regimen was evaluated in CLL patients who had not received prior treatment. Among 224 patients in this trial, the CR rate was 70%, and the ORR was 95%. Pretreatment characteristics were significantly associated with a lower probability of CR (p < 0.05), including advanced Rai and Binet staging, older age, higher white cell count, lower platelet count, higher serum β2-microglobulin level, >50% cellularity on bone marrow biopsy, and spleen enlargement >5 cm below the left costal margin. Flow cytometry assessment of residual disease in bone marrow aspirates was performed in 207 patients, with 67% of patients showing <1% co-expression of CD5 and CD19 cells, considered "flow cytometry CR." The most common complications of this regimen were bone marrow suppression and infectious complications. The latest results of this trial showed a 6-year overall survival rate and failure-free survival rate of 77% and 51%, respectively, with a median progression time of 80 months.
The CLL8 phase III trial was the first large randomized trial comparing the efficacy of FC and FCR in newly diagnosed CLL patients. In an open-label manner, 817 patients were randomly assigned to one of the regimens. The recently published updated results of this trial showed a median follow-up time of 5.9 years, with PFS of 56.8 months for FCR and 32.9 months for FC (hazard ratio (HR), 0.59; 95% confidence interval (CI), 0.50–0.69, p < 0.001). Furthermore, OS superiority was significant: FCR group not reached OS, while FC group was 86 months (HR, 0.68; 95% CI, 0.54–0.89, p = 0.001). The main toxicities associated with this therapy were bone marrow suppression (potentially prolonged) and infectious complications.
Drug interactions may alter the way drugs work or increase the risk of serious side effects. This document does not contain all possible drug interactions. Keep a list of all the products you use (including prescription/nonprescription drugs and herbal products) and share it with your doctor and pharmacist. Do not start, stop, or change the dosage of any medicine without your doctor's approval.
Some products that may interact with this drug include: "blood thinners" (such as warfarin, heparin), live vaccines (such as nasal flu vaccine, oral typhoid/paratyphoid vaccine), sulfinpyrazone, other drugs that weaken the immune system/increase infection risk (such as natalizumab, rituximab), salicylates/nonsteroidal anti-inflammatory drugs (such as aspirin, ibuprofen, naproxen).
Check all prescription and nonprescription medicine labels carefully, as many labels contain analgesics/antipyretics (nonsteroidal anti-inflammatory drugs such as ibuprofen, naproxen, and aspirin), which may increase bleeding risk. If the medicine prescribed by your doctor is for preventing heart attacks or strokes, continue taking low-dose aspirin (usually 81-162 milligrams per day). Consult your doctor or pharmacist for details.
Research on the combined use of fludarabine and cyclophosphamide indicates significant efficacy in treating chronic lymphocytic leukemia (CLL). Many clinical trials and research findings support its advantages in improving response rates, extending progression-free survival, and overall survival. However, these studies also highlight the potential side effects and risks associated with combination therapy. Therefore, in practical clinical application, doctors need to weigh the benefits and risks and tailor the most suitable treatment plan for individual patients. In the future, with more research and the emergence of new therapies, the potential of combining fludarabine and cyclophosphamide will be further explored, bringing more hope and choices to patients.
[1]https://go.drugbank.com/drugs/DB01073
[2]https://go.drugbank.com/drugs/DB00531
[3]https://link.springer.com/article/10.1007/s12185-022-03488-5
[4]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5305006/
[5]https://www.ncbi.nlm.nih.gov/books/NBK304336/
[6]https://www.webmd.com/drugs/2/drug-8784/fludarabine-intravenous/details
[7]https://www.cancerresearchuk.org/about-cancer/treatment/drugs/fcr
 |
 |
 |