This article extensively investigates the decomposition of hexamethylenetetramine (HMTA), aiming to gain a deep understanding of its reaction pathways and product formation under various conditions.
Hexamethylenetetramine (HMTA) is a well-known nitrogen heterocycle characterized by a cage-like structure reminiscent of adamantane. This multifunctional compound finds numerous scientific and technological applications, particularly noted for its ability to form metal complexes as highlighted in several reports. Recently, interest in HMTA's applications in materials science has surged, establishing it as an ideal component for manufacturing ZnO microflowers and various nanomaterial categories. Specific applications of HMTA across various chemical fields include:
Hexamethylenetetramine is a significant chemical product used as a curing agent for phenol-formaldehyde plastics, a catalyst for amino plastics, a promoter for rubber vulcanization (Promoter H), a shrink-proof agent in textile production, a preservative in food processing, a diuretic in medicine; in agriculture, it can be used as an insecticide; in military, it can be used as an absorber for phosgene in gas masks; after nitration treatment, it is also an important explosive.
Hexamethylenetetramine undergoes decomposition under specific conditions. Depending on the factors involved, this decomposition can follow different pathways.
The decomposition of HMTA depends on temperature and surrounding environment. There are two main scenarios:
At high temperatures (approximately 200-800°C), HMTA primarily decomposes into ammonia (NH3) and formaldehyde (CH2O) through a series of complex reactions. The exact mechanism is still under investigation but likely involves the cleavage of C-N bonds within the HMTA cage structure.
In acidic environments, HMTA decomposes via different pathways. The specifics depend on the particular acid used but generally involve protonation of HMTA molecules followed by rearrangement and release of ammonia and formaldehyde.
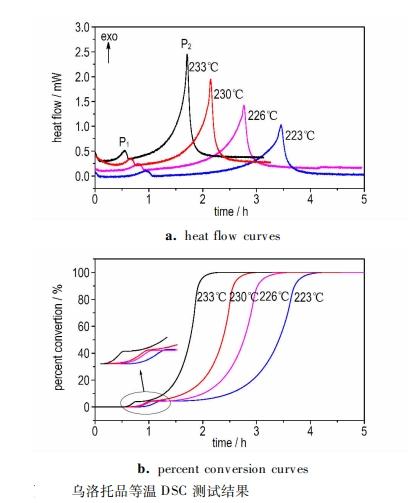
Peng Haoliang et al. obtained the thermal decomposition kinetic parameters of HMTA by testing its thermal decomposition process using differential scanning calorimetry (DSC) and adiabatic accelerating calorimetry (ARC). The DSC results indicate that the thermal decomposition of HMTA is a coupled exothermic process, with two exothermic peaks in its isothermal test, corresponding to the apparent activation energies of 150 kJ·mol-1, and the maximum temperature rise rate to time TD24 of 216.26℃ was calculated using AKTS software. The ARC test results indicate that the initial decomposition temperature of HMTA was 230.28℃, TD24 was 212.5℃, which is basically consistent with the prediction results based on isothermal DSC data (216.26℃).
Temperature plays a crucial role in HMTA decomposition. At low temperatures (around room temperature), the decomposition process is slow, primarily releasing ammonia and formaldehyde through hydrolysis. As the temperature increases, decomposition accelerates. Within the range of 200-300℃, ammonia and formaldehyde remain the primary products. At higher temperatures (300-800℃), the decomposition pathway changes. The release of ammonia decreases while the formation of toxic products such as hydrogen cyanide (HCN) increases.
Acidity and alkalinity: HMTA decomposition may be influenced by the surrounding environment's pH value. Compared to thermal decomposition, acids can promote HMTA decomposition through different pathways.
Presence of oxidants: Strong oxidants can accelerate HMTA decomposition, potentially leading to the formation of more hazardous products.
Certain substances can act as catalysts, significantly affecting the rate and products of HMTA decomposition. Most reactions involving HMTA use stoichiometric or excess acids, which hinder reaction sustainability. Preeti Devi et al. reported the first example of ruthenium-catalyzed HMTA decomposition at room temperature, supported by detailed mechanism studies using thermogravimetric/differential thermal analysis, variable temperature NMR, UV-visible spectroscopy, and density functional theory. The mechanism studies also involved a comparison of the decomposition of HMTA, protonated HMTA, [RuCl3(HMTA)], and [FeCl3(HMTA)], indicating that [RuCl3(HMTA)] decomposes at the lowest temperature and has the lowest HOMO-LUMO band gap of 2.66 eV. Ruthenium-induced HMTA decomposition has been successfully used as a tool to increase the sustainability of the Sommelet reaction, as it uses simple RuCl3·nH2O as a catalyst at concentrations as low as 0.5 mol%, in aqueous media. Even for highly challenging substrates, i.e., aliphatic aldehydes and substrates with electron-withdrawing substituents, the developed method exhibits high selectivity and efficiency. The results of this study represent the first use of ruthenium in the Sommelet reaction and provide a possible platform for improving the sustainability of all reactions involving HMTA as a reactant/reagent.
A large amount of hexamine is used in casting, tires and rubber, phenolic resin industry and other different applications. It is reported that excessive exposure to solid hexamine or its vapors can cause dermatitis and respiratory allergies. Although Ames tests of hexamine are positive, most animal studies indicate that hexamine has a very low genetic risk, even at very high doses. The effectiveness of hexamine as an antimicrobial agent is attributed to its slow hydrolysis into ammonia and formaldehyde. In industries using hexamine at high temperatures, concerns have arisen about the emission of HCN, NH3, CO, CO2, nitrogen oxides and formaldehyde. Studies unanimously believe that the characteristics of hexamine thermal decomposition increase with temperature, increase HCN and reduce NH3 emissions between 300 and 800°C. Reported at temperatures of 200-300°C, hexamine decomposition mainly produces ammonia and formaldehyde.
In the report of Jinjun Lin et al., when HMTA sublimes at high temperatures, it decomposes into various compounds such as trimethylamine ((CH3)3N), dimethylamine ((CH3)2NH), methylamine ((CH3NH2), ethylene imine ((CH2)2NH), acrylonitrile (CH3CH2CN) and acetonitrile (CH3CN). These heavier compounds (or HMTA itself) can further (directly) decompose into lighter gases such as methane (CH4), ammonia (NH3), hydrogen (H2) and nitrogen (N2), which are basic gas sources for manufacturing NG films. Since precursor gases can be controlled by using appropriate sublimation temperatures, the use of this single-source solid precursor can achieve relatively uniform growth of NG single crystals.
Slow release of antimicrobials. HMTA slow hydrolysis (and water hydrolysis) to release ammonia and formaldehyde. This property allows it to serve as a sustained-release source for these antimicrobials in certain urinary tract medications. Understanding the decomposition products helps ensure controlled release and minimizes potential side effects.
HMTA decomposes at high temperatures, releasing formaldehyde. This formaldehyde can serve as a curing agent for various resins, such as phenol-formaldehyde resins used in adhesives and laminates. Understanding the decomposition temperature allows precise control of the curing process, optimizing product quality and consistency.
HMTA can be used as a foaming agent for certain foams. After decomposition, it generates gas, causing the material to expand and form lightweight foam structures. Understanding the decomposition products ensures that the gas produced is safe and does not compromise foam characteristics.
Under specific conditions, HMTA decomposition can be rapid and exothermic (heat releasing). This may pose an explosion hazard. Understanding the decomposition behavior of HMTA is crucial for safe storage, handling, and transportation in industries using HMTA.
At high temperatures, HMTA decomposition produces highly toxic gases such as hydrogen cyanide (HCN). Understanding the temperature threshold for decomposition helps prevent HCN formation and ensures worker safety.
Research on hexamethylenetetramine decomposition provides us with an in-depth understanding of its stability and reaction mechanisms. Despite making some progress, many unresolved issues remain, especially regarding the kinetics of decomposition reactions and product analysis under different environmental conditions. Future research will continue to explore the mechanisms of hexamethylenetetramine decomposition and its potential impacts on the environment and health, contributing to better understanding and management of the compound's applications and effects.
[1] https://pubs.rsc.org/en/content/articlelanding/2021/ob/d1ob01522b
[2] https://pubmed.ncbi.nlm.nih.gov/2688388/
[3] https://www.sciencedirect.com/science/article/abs/pii/S0169433218314387
[4] https://pubmed.ncbi.nlm.nih.gov/38482798/
[5] Peng Haoliang, Chen Liping, Lu Guibin, et al. Thermal decomposition kinetics of hexamethylenetetramine[J]. Energetic Materials, 2016, 24(5): 497-502. DOI: 10.11943/j.issn.1006-9941.2016.05.012.
 |
 |
 |