Hydroxypropyl beta cyclodextrin (HPCD) is an important chemical compound with diverse applications. Its unique chemical structure and biocompatibility have made it a focal point in research across pharmaceuticals, food, and other industries.
Hydroxypropyl beta cyclodextrin (HPCD) consists of seven sugar molecules bonded together to form a cyclic oligosaccharide. It features a hydrophobic cavity at its center capable of capturing cholesterol and lipids. Over the past century, it has been widely used as a complexing agent in the food, deodorant, and pharmaceutical industries, recognized as Generally Recognized As Safe (GRAS).
As an active ingredient, HPCD can capture and clear cholesterol and lipids within cells, which can otherwise damage organs such as the kidneys, brain, and liver.
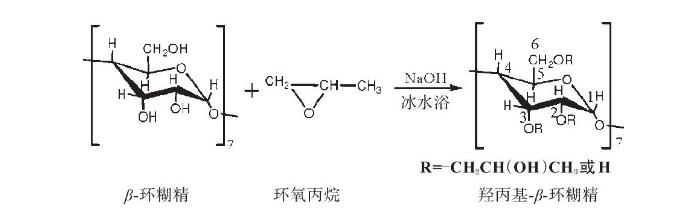
The molecular formula of HPCD is C54H102O39, with hydroxypropyl beta cyclodextrin molecular weight of 1375.36. HPCD is synthesized by the reaction of beta-cyclodextrin with epichlorohydrin under alkaline conditions, utilizing a bimolecular nucleophilic substitution (SN2) mechanism. In the presence of a strong base, epichlorohydrin attacks the oxygen atom with the least substituted carbon in the cyclodextrin ring, primarily at the 6-position. This reaction results in a mixture of products due to the many hydroxyl groups present on beta-cyclodextrin, influencing the hydroxypropyl beta cyclodextrin structure and properties of HPCD.
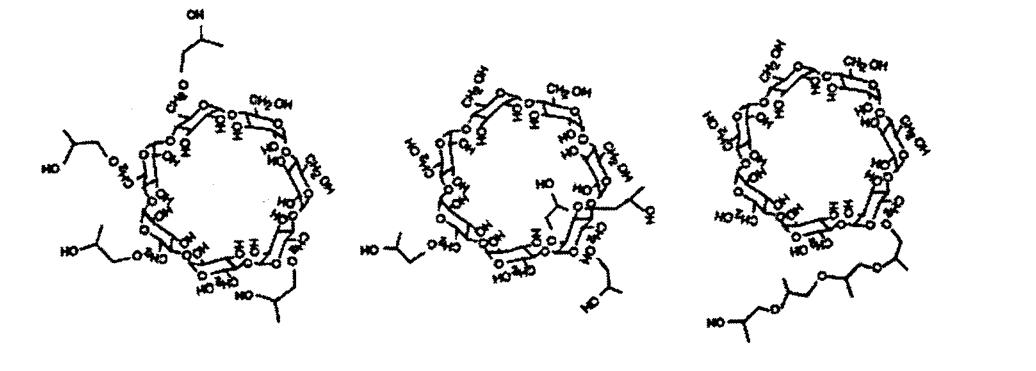
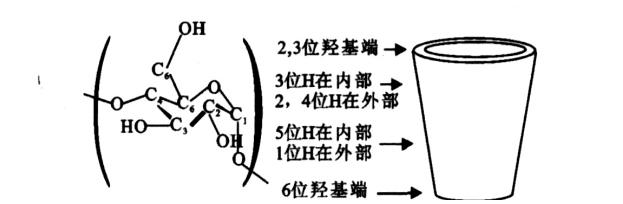
Long believed to have a rigid structure, recent studies suggest that cyclodextrins and their derivatives exhibit relative flexibility, facilitating complex formation through non-covalent interactions. This flexibility enhances the formation capacity of complexes and reaction kinetics in both solution and solid states.
In pharmaceuticals, HPCD serves as an excipient to enhance drug delivery. Many drugs have poor solubility in water, hindering absorption in the body. HPCD forms complexes with these drugs, increasing their solubility and thereby improving their efficacy. This not only enhances the delivery of existing drugs but also facilitates the development of formulations for poorly soluble drugs.
The ability of HPCD to capture and slowly release ingredients enhances the stability and efficacy of cosmetics. For example, it can encapsulate fragrances, prolonging their lifespan and preventing rapid evaporation. Moreover, HPCD improves the delivery of active ingredients in skincare products, enhancing penetration and potentially boosting their effectiveness.
HPCD can mask bitterness or unpleasant aftertastes, thereby improving the taste of food products. Additionally, it enhances the stability of food additives (e.g., vitamins and antioxidants), prolonging shelf life and ensuring efficacy throughout consumption.
In certain industrial processes, HPCD is used to remove unwanted components from mixtures or control the release of chemicals during reactions.
One of the primary advantages of HPCD is its ability to improve the solubility and stability of poorly soluble drugs. These drugs often have low water solubility, which hampers their absorption in the body. HPCD acts as a host molecule, forming inclusion complexes with drug molecules. This complexation increases the drug's solubility in water, enhancing absorption and potentially increasing bioavailability. Furthermore, the cyclic structure of HPCD provides protection to drug molecules, enhancing their stability and preventing degradation during storage or in vivo.
By enhancing the solubility and stability of drugs, HPCD significantly improves their bioavailability—the proportion of a drug that enters circulation and reaches the target site to exert its therapeutic effect. Poorly soluble drugs often suffer from limited dissolution and reduced availability for absorption. HPCD helps overcome this challenge by promoting better dissolution and thereby increasing the amount of drug entering the bloodstream and reaching the target site. This translates into more effective therapeutic outcomes and potentially lowers the required drug dosage.
Beyond its role as an excipient, HPCD itself possesses properties that contribute to enhancing drug efficacy. In some cases, these properties may synergistically interact with active drug ingredients, potentially boosting overall therapeutic effectiveness.
Hydroxypropyl beta cyclodextrin can be purchased from various suppliers and manufacturers. Due to its potential applications in research and medicine, sourcing from reputable suppliers is crucial. Several online retailers offer HPCD, but it is essential to check product specifications and intended uses before purchase. Well-established companies provide contact information on their websites for further inquiries.
As a functional nanomaterial, hydroxypropyl beta cyclodextrin demonstrates extensive application prospects and significant scientific value. With ongoing research into its properties and applications, HPCD is poised to play an increasingly important role in relevant fields, contributing to human health and environmental protection.
[1] Cai, S., Huang, H., & Chen, L. (2008). Development of Hydroxypropyl Beta Cyclodextrin. China Pharmacy, (10), 78-79.
[2] Yuan, C. (2008). Preparation, Properties, and Applications of Hydroxypropyl Beta Cyclodextrin (Master's thesis). Jiangnan University.
[3] ZyVersa Therapeutics. About Hydroxypropyl Beta Cyclodextrin. Retrieved from https://www.zyversa.com/pipeline/about-hydroxypropyl-beta-cyclodextrin
[4] MDPI. (2021). Hydroxypropyl Beta Cyclodextrin: Applications and Development. Retrieved from https://www.mdpi.com/2076-3921/12/3/552
[5] MDPI. (2022). Recent Advances in the Application of Hydroxypropyl Beta Cyclodextrin. Retrieved from https://www.mdpi.com/2218-0532/87/4/33
 |
 |
 |