-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

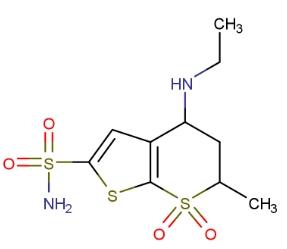
Dorzolamide is a commonly used medication primarily for treating eye conditions such as glaucoma and high eye pressure. Widely applied in ophthalmology, dorzolamide has shown promising efficacy in many patients. This article explores the mechanism of action, uses, and significance of dorzolamide in ophthalmic treatment.
Dorzolamide, categorized as a carbonic anhydrase inhibitor medication, works by reducing the production of fluid inside the eye, aiding in lowering eye pressure and alleviating symptoms and risks in glaucoma patients. Typically administered in the form of eye drops, dorzolamide allows for direct application to the eye, facilitating therapeutic effects. Dorzolamide acts by reducing the production of fluid inside the eye, which, in turn, helps to lower pressure and protect the optic nerve from damage. This aids in preserving vision and preventing further complications of glaucoma.
Carbonic anhydrase (CA) is an enzyme present in many tissues of the body, including the eyes. It catalyzes reversible reactions involving the hydration of carbon dioxide and the dehydration of carbonic acid. In humans, carbonic anhydrase exists in various isoforms, with the most active being carbonic anhydrase II (CA-II), primarily present in red blood cells (RBCs) but also found in other tissues. Inhibiting carbonic anhydrase in the ciliary processes of the eye reduces aqueous humor secretion, possibly by slowing the formation of bicarbonate ions, subsequently reducing sodium and fluid transport. The result is a decrease in intraocular pressure (IOP).
Dorzolamide belongs to a class of drugs called carbonic anhydrase inhibitors. It works by blocking an enzyme called carbonic anhydrase in the eye. This enzyme functions in producing a fluid called aqueous humor in the front part of the eye. By inhibiting carbonic anhydrase, dorzolamide reduces the amount of fluid produced in the eye. With decreased fluid production, intraocular pressure decreases. This helps prevent worsening of glaucoma and damage to your vision.
After topical administration, dorzolamide eye drops can reduce elevated intraocular pressure. Elevated intraocular pressure is a major risk factor for optic nerve damage and the pathogenesis of glaucomatous visual field defects.
Dorzolamide and timolol in combination are used to treat eye conditions including glaucoma and high intraocular pressure, where increased pressure can lead to gradual vision loss. Dorzolamide and timolol are used in patients who do not respond to another drug for eye conditions. Dorzolamide belongs to a class of drugs called topical carbonic anhydrase inhibitors. Timolol belongs to a class of drugs called topical beta-adrenergic receptor blockers. Dorzolamide and timolol reduce eye pressure by reducing the production of natural fluids in the eye.
Both dorzolamide and latanoprost are medications used to lower glaucoma and high intraocular pressure, but they achieve this through different mechanisms: dorzolamide, this medication belongs to a class of drugs called carbonic anhydrase inhibitors. It directly reduces the production of aqueous humor in the eye. By reducing the production of fluid, dorzolamide lowers IOP; latanoprost belongs to the class of prostaglandin analogs. Its mechanism of action is to increase the outflow of aqueous humor through another pathway in the eye, the uveoscleral outflow pathway. Increased drainage leads to decreased IOP.
Nicola Orzalesi et al. compared the effects of 0.5% timolol, 0.005% latanoprost, and dorzolamide on the 24-hour intraocular pressure (IOP) reduction in patients with primary open-angle glaucoma (POAG) or ocular hypertension (OHT). Methods: In this crossover trial, 20 POAG (n = 10) or OHT (n = 10) patients received treatment with timolol, latanoprost, and dorzolamide for 1 month. The treatment sequence was randomized. All patients underwent four 24-hour IOP curve measurements: at baseline and after each month of treatment. Patients were admitted to the hospital, and eye pressure was measured by two trained evaluators who were blinded to the treatment allocation. Measurements were taken at 3 a.m., 6 a.m., and 9 a.m., as well as at noon, 3 p.m., 6 p.m., and 9 p.m., and midnight using a handheld electronic tonometer (TonoPen XL; Bio-Rad, Glendale, California), with patients in supine and sitting positions, and with a Goldmann tonometer. (Haag-Streit, Bern, Switzerland), with patients seated at the slit lamp. Blood pressure was also recorded. Group differences were tested for significance using parameter variance analysis. The same procedure was also used to record the diurnal IOP curves of a small group of untreated healthy young participants. To compare the diurnal and nocturnal IOP rhythms between the POAG-OHT group and the control group, the final endpoint was calculated for each subject.
Except for timolol at 3 a.m., all drugs significantly reduced IOP compared to baseline when considering Goldmann sitting values. Latanoprost was more effective than timolol in reducing IOP at 3 a.m., 6 a.m., and 9 a.m. (P = 0.03), noon (P = 0.01), 9 p.m., and midnight (P = 0.05) compared to dorzolamide. (P = 0.03) and 3 p.m. and 6 p.m. (P = 0.04). At 3 p.m., timolol was more effective than dorzolamide (P = 0.05), while dorzolamide was more effective than timolol at midnight and 3 a.m. (P = 0.05). An auxiliary finding of this study was that the pattern of eye pressure curves in healthy subjects differed from that in patients with eye diseases.
The graph below shows the Goldmann tonometer readings of the baseline, timolol, latanoprost, and dorzolamide diurnal and nocturnal rhythm curves. All drugs significantly reduced eye pressure at all time points compared to baseline, except for timolol at 3 a.m. The mean eye pressure was 22.7 ± 1.8 mm Hg at baseline, 18.7 ± 0.9 mm Hg for timolol, 16.3 ± 0.6 for latanoprost, and 19.3 ± 1.7 for dorzolamide. When comparing latanoprost to timolol (P = 0.001) and dorzolamide (P = 0.001), the average IOP difference was statistically significant. There was no statistically significant difference in mean eye pressure between timolol and dorzolamide.
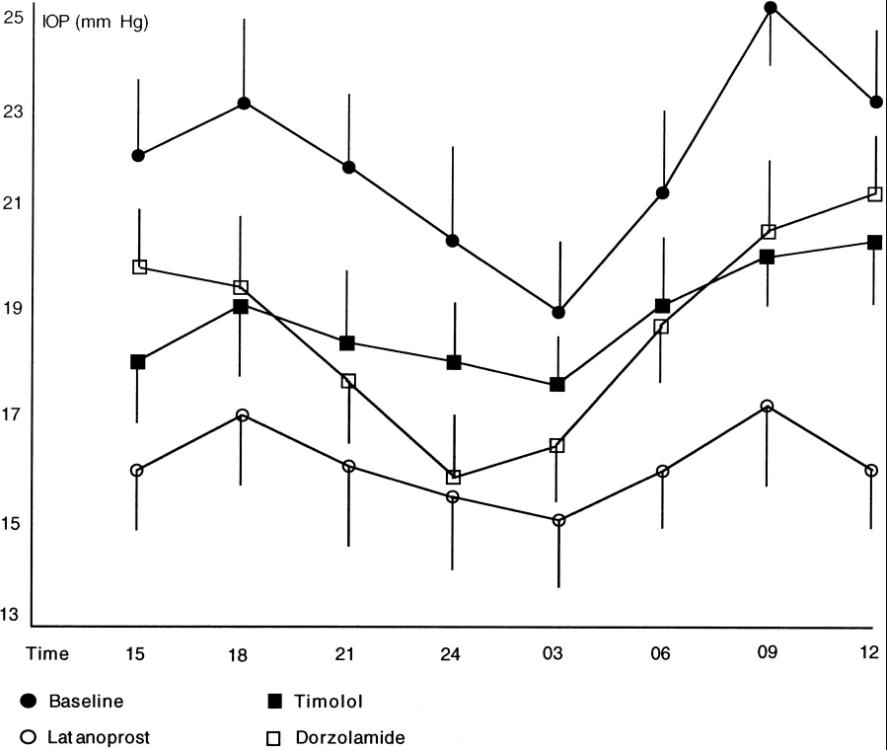
In summary, latanoprost appears to reduce the diurnal and nocturnal rhythm of eye pressure, while timolol appears to be less effective at night. The effect of dorzolamide is less than that of latanoprost but significantly reduces nocturnal eye pressure. The reason for the differences in eye pressure curve patterns in healthy subjects is currently unclear and warrants further investigation.
There is inadequate and well-controlled studies in pregnant women using Dorzolamide hydrochloride ophthalmic solution. Dorzolamide, administered orally at 2.5 mg/kg/day (37 times the clinical exposure), induced vertebral malformations in fetuses of rabbits. Dorzolamide administered to rabbits during organogenesis at oral doses of up to 1 mg/kg/day (15 times the clinical exposure) did not induce malformations. Administration of Dorzolamide hydrochloride to rats during late gestation and lactation resulted in offspring growth retardation at an oral dose of 7.5 mg/kg/day (52 times the clinical exposure). Growth was not delayed at 1 mg/kg/day (8.0 times the clinical exposure).
The background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
There are no data on the presence of Dorzolamide hydrochloride ophthalmic solution in human milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for Dorzolamide hydrochloride ophthalmic solution and any potential adverse effects on the breastfed child from Dorzolamide hydrochloride ophthalmic solution. Dorzolamide is present in the milk of lactating rats.
Lactating rats were administered oral Dorzolamide hydrochloride at 7.5 mg/kg/day; Dorzolamide and its N-deethylated metabolite were detected in milk.
The safety and effectiveness of Dorzolamide hydrochloride ophthalmic solution have been established in a multicenter, double-blind, active-controlled trial in pediatric patients over a 3-month period.
No overall differences in safety or effectiveness were observed between elderly and younger patients.
Dorzolamide has not been studied in patients with severe renal impairment (CrCl < 30 mL/min). Because Dorzolamide and its metabolites are primarily excreted by the kidney, use of Dorzolamide hydrochloride ophthalmic solution in these patients is not recommended.
Dorzolamide has not been studied in patients with hepatic impairment and caution should be exercised in these patients.
Before using dorzolamide, inform your doctor or pharmacist if you are allergic to it or if you have any other allergies. This product may contain inactive ingredients (such as preservative benzalkonium chloride), which can cause allergic reactions or other problems. Consult your pharmacist for more details.
Before using this medication, inform your doctor or pharmacist about your medical history, especially of: kidney disease, kidney stones, liver disease.
If you have an eye infection or injury, or if you are undergoing eye surgery, consult your doctor to see if you should continue using your current bottle of dorzolamide. You may be advised to start using a new bottle.
Before surgery, inform your doctor or dentist about all the products you use (including prescription drugs, nonprescription drugs, and herbal products).
Your vision may temporarily become blurred or unstable after using this medication. Do not drive, use machinery, or do any activity that requires clear vision until you can do it safely.
Eye stinging, burning, itching, and bitterness. The most common adverse reactions associated with dorzolamide are bitterness and transient local burning or stinging. In a large study, conjunctivitis was the most common reason for discontinuation of dorzolamide. It causes shallowing of the anterior chamber and transient myopia. As a second-generation carbonic anhydrase inhibitor, dorzolamide avoids the systemic effects associated with first-generation carbonic anhydrase inhibitors (such as acetazolamide, methazolamide, and dichlorphenamide).
Dorzolamide, as a carbonic anhydrase inhibitor medication, plays a significant role in the treatment of glaucoma and high eye pressure. By reducing the production of intraocular fluid, it helps to lower eye pressure, alleviate symptoms, and reduce the risk of disease progression. However, the use of dorzolamide may also cause some side effects and adverse reactions, so caution and close attention to individual responses are warranted during use. If you have any questions or need further information about dorzolamide, it is advisable to consult a professional ophthalmologist to ensure the effectiveness of your treatment and the health of your eyes.
[1] Orzalesi N, Rossetti L, Invernizzi T, et al. Effect of timolol, latanoprost, and dorzolamide on circadian IOP in glaucoma or ocular hypertension[J]. Investigative Ophthalmology & Visual Science, 2000, 41(9): 2566-2573.
[2] Balfour J A, Wilde M I. Dorzolamide: a review of its pharmacology and therapeutic potential in the management of glaucoma and ocular hypertension[J]. Drugs & aging, 1997, 10: 384-403.
[3] https://en.wikipedia.org/wiki/Dorzolamide
[4] https://medlineplus.gov/druginfo/meds/a602022.html
[5] https://www.drugs.com/pro/dorzolamide.html
[6] https://www.webmd.com/drugs/2/drug-6076/dorzolamide-ophthalmic-eye/details
 |
 |
 |