-
We detected your language preference as English. Would you like to switch to the English version for a better experience?
Switch to English
Stay here

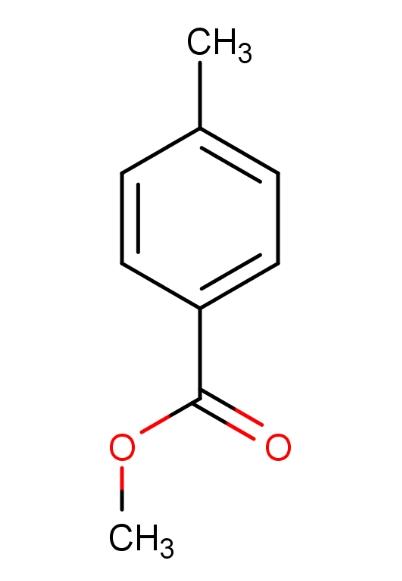
Methyl 4-methylbenzoate, with CAS number 99-75-2 and molecular formula C9H10O2, is a crystalline solid ranging from colorless to white, emitting a pleasant aromatic scent. This ester compound is naturally found in certain fruits and can also be produced synthetically, with broad applications across various industrial fields.
Methyl 4 methylbenzoate uses
Methyl 4-methylbenzoate is highly valued in the fragrance industry for its versatile and enchanting aroma. It not only adds rich layers to ylang-ylang fragrances but also imparts a strong floral scent to soaps and detergents. Additionally, its low cost makes it an excellent choice for masking industrial raw material odors. According to the RIFM fragrance ingredient safety assessment, the maximum acceptable concentration of methyl 4-methylbenzoate in finished products is as follows:

Methyl 4-methylbenzoate is a versatile compound with significant uses in chemical synthesis.
In Laboratory and Industrial Chemical Synthesis
This chemical is a valuable starting material for producing other compounds. Its reactive functional groups allow for various chemical transformations, making it a cornerstone in the development of new molecules. Methyl 4-methylbenzoate can be hydrolyzed to produce 4-toluic acid, which has wide applications across multiple industries. Additionally, reduction of the ester group can yield 4-methylbenzyl alcohol, a crucial intermediate used in drug synthesis and the production of other fine chemicals.
Benefits of Enhanced Reaction Efficiency
Using methyl 4-methylbenzoate typically improves reaction efficiency. Its specific chemical properties can facilitate the desired transformations, shorten reaction times, and increase product yields. Reduction of the ester group results in 4-methylbenzyl alcohol, a valuable intermediate in synthesizing pharmaceuticals and other fine chemicals.
In the pharmaceutical industry, this compound is often used as an intermediate in synthesizing various drugs. Its unique chemical structure allows for further modification, providing extensive potential in drug design and development.
Reference Standards
Methyl 4-methylbenzoate is commonly used as a reference standard in analytical techniques such as gas chromatography (GC) and liquid chromatography (LC). Its precise retention time and response factor provide a reliable basis for quantitative analysis.
Impurity Identification and Removal
In organic synthesis, methyl 4-methylbenzoate may appear as a byproduct. By studying its formation mechanisms and properties within reaction systems, efficient impurity removal methods can be developed, enhancing the purity of the target product.
Methyl 4-methylbenzoate's relatively simple structure and representative ester characteristics make it a model compound for studying various organic reactions. Detailed studies of its reactivity can reveal the effects of substituents on the reaction activity and selectivity of the aromatic ring.
Solution Properties
The solubility of methyl 4-methylbenzoate in different solvents is closely related to its molecular structure and solvent polarity. Systematic studies of its solubility behavior can provide insights into intermolecular interactions and solvation effects.
Thermodynamic Properties
Thermodynamic properties such as heat capacity and enthalpy changes of methyl 4-methylbenzoate can offer theoretical insights into its phase behavior under varying temperatures and pressures.
Methyl 4-methylbenzoate and its derivatives can be used as monomers or intermediates for synthesizing new functional materials. For example, by introducing different substituents, the physical and chemical properties of materials can be tuned, offering potential applications in optical and electrical fields.
[1]Chen X, Johnson R M, Li B. A Permissive Amide N-Methyltransferase for Dithiolopyrrolones[J]. ACS catalysis, 2023, 13(3): 1899-1905.
[2]https://www.ncbi.nlm.nih.gov/books/NBK592503/
[3]Api A M, Belsito D, Botelho D, et al. RIFM fragrance ingredient safety assessment, methyl p-methylbenzoate, CAS Registry Number 99-75-2[J]. 2022.
[4]https://en.wikipedia.org/wiki/Methyl_p-toluate
[5]https://webbook.nist.gov/
 |
 |